Electrode
Electrode is a conductor through which electricity enters or leaves an object, substance, or region. They are used in a wide range of applications, including but not limited to, electronics, chemistry, and medicine.
Overview[edit]
An Electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). The word was coined by William Whewell at the request of the scientist Michael Faraday from two Greek words: elektron, meaning amber (from which the word electricity is derived), and hodos, a way.
Types of Electrodes[edit]
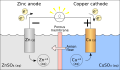
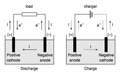
There are two types of electrodes: Anode and Cathode. The anode is positively charged, while the cathode is negatively charged.
Anode[edit]
The Anode is the electrode where oxidation (loss of electrons) takes place. In a galvanic cell, it is the negative electrode, as it is the electrode that provides electrons. In an electrolytic cell, it is the positive electrode.
Cathode[edit]
The Cathode is the electrode where reduction (gain of electrons) takes place. In a galvanic cell, it is the positive electrode, as it is the electrode that accepts electrons. In an electrolytic cell, it is the negative electrode.
Electrodes in Medicine[edit]
In medicine, electrodes are used in numerous ways, including in ECG machines to monitor heart activity, in EEG machines to monitor brain activity, and in ECT for treating mental disorders.
See Also[edit]
- Electrolysis
- Electrochemical cell
- Battery
- Fuel cell
- Electrode potential
- Standard electrode potential
References[edit]
-
Arc welding electrodes and electrode holder
-
Galvanic cell with no cation flow
-
Batteries
-
Rechargeable batteries
-
Battery discharge and charge
-
Hush adiabatic electron transfer model parameters
-
Redox flow battery
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian