Dithionite
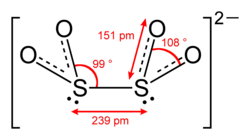

Dithionite is a chemical compound with the formula S2O4^2-. It is a reducing agent, commonly encountered as the sodium salt, sodium dithionite (Na2S2O4). Dithionite is used in a variety of applications, ranging from the bleaching of paper pulp to the reduction of metal ions in solution. Its reducing power, combined with its relatively low toxicity, makes it a valuable chemical in both industrial and laboratory settings.
Properties[edit]
Dithionite is a strong reducing agent, capable of donating electrons to various acceptors. It is stable under alkaline conditions but decomposes rapidly in acidic solutions to form sulfur dioxide and sulfide. This instability in acidic environments limits its use in certain applications. Dithionite is also sensitive to oxygen and will slowly oxidize upon exposure to air, necessitating storage under inert or reducing conditions.
Production[edit]
The industrial production of dithionite typically involves the reduction of sulfur dioxide with zinc or by electrolytic methods. The zinc process involves reacting sulfur dioxide with zinc dust and sodium hydroxide, leading to the formation of sodium dithionite and zinc oxide as by-products. The electrolytic method, on the other hand, involves the direct reduction of sulfur dioxide in a sodium hydroxide solution.
Applications[edit]
Bleaching Agent[edit]
One of the primary uses of dithionite is as a bleaching agent in the paper and textile industries. In the paper industry, it is used to remove lignin from wood pulp, resulting in a brighter pulp. In the textile industry, dithionite is used to bleach cotton and linen, removing natural pigments and preparing the fabrics for dyeing.
Metal Reduction[edit]
Dithionite is also employed in the reduction of metal ions, particularly in the recovery of precious metals. It can reduce metal ions in solution to their metallic forms, facilitating their recovery from ores and waste streams.
Chemical Synthesis[edit]
In organic and inorganic chemistry, dithionite is used as a reducing agent in various synthesis reactions. It can reduce nitro compounds to amines, ketones to alcohols, and disulfides to thiols, among other applications.
Safety[edit]
While dithionite is not highly toxic, it requires careful handling due to its reducing power and sensitivity to acid and oxygen. Exposure to dithionite can cause irritation to the skin, eyes, and respiratory system. Proper personal protective equipment, such as gloves and goggles, should be worn when handling dithionite.
Environmental Impact[edit]
The use of dithionite in industrial processes can lead to the release of sulfur compounds into the environment. Proper waste treatment and disposal practices are necessary to minimize its environmental impact.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
