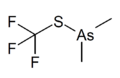
Dimethyl(trifluoromethylthio)arsine
Dimethyl(trifluoromethylthio)arsine is a chemical compound with the formula C_2H_6AsSCF_3. This compound belongs to the class of organometallic compounds, specifically an arsine derivative, where arsenic is covalently bonded to carbon and sulfur atoms. The presence of the trifluoromethylthio group (SCF_3) introduces significant electronegativity due to the fluorine atoms, affecting the compound's reactivity and physical properties.
Properties and Synthesis[edit]
Dimethyl(trifluoromethylthio)arsine is characterized by its unique structural framework, which combines the elements of arsenic, sulfur, and fluorine. The synthesis of this compound typically involves the reaction of dimethylarsine (Me_2AsH) with trifluoromethanethiol (CF_3SH), under controlled conditions. This process requires careful handling due to the toxic and reactive nature of arsenic compounds.
Applications[edit]
The applications of Dimethyl(trifluoromethylthio)arsine are primarily in the field of organic synthesis and catalysis. Its ability to introduce the trifluoromethylthio group into organic molecules makes it a valuable reagent in the synthesis of pharmaceuticals and agrochemicals. Furthermore, its organoarsenic framework can be exploited in catalytic cycles, particularly in the formation of carbon-sulfur and carbon-arsenic bonds.
Safety and Handling[edit]
Handling of Dimethyl(trifluoromethylthio)arsine requires strict safety measures due to its toxicity and potential for releasing toxic arsenic fumes upon decomposition. Appropriate personal protective equipment (PPE) and ventilation are essential when working with this compound. Environmental regulations also dictate the disposal and containment measures to prevent arsenic contamination.
```
Dimethyl(trifluoromethylthio)arsine[edit]
-
Structure of Dimethyl(trifluoromethylthio)arsine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian