Diiodomethane
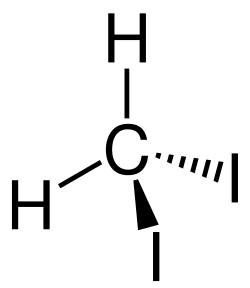
Diiodomethane is a chemical compound with the formula CH2I2. It is a dense, colorless, volatile liquid at room temperature and pressure, known for its use in various organic synthesis processes and as a high-density solvent in mineral separation techniques. Diiodomethane is notable for its application in the Nuclear Magnetic Resonance (NMR) spectroscopy and in the determination of the density of minerals by the heavy liquid separation method.
Properties[edit]
Diiodomethane has a molecular weight of 267.84 g/mol and a boiling point of 181 °C. It is slightly soluble in water, but more soluble in organic solvents such as ethanol, diethyl ether, and acetone. The high density of diiodomethane, approximately 3.325 g/cm^3, makes it useful for the separation of minerals and other substances based on density differences.
Synthesis[edit]
Diiodomethane can be synthesized through several methods, including the direct reaction of iodine with methane or methanol in the presence of red phosphorus or by the reaction of iodine with methylene chloride in the presence of an oxidizing agent like nitric acid.
Applications[edit]
Organic Synthesis[edit]
In organic chemistry, diiodomethane is used as a source of methylene groups in various synthesis reactions. It participates in the Wittig reaction and can act as a methylene donor in the formation of cyclopropanes.
Density Gradient Separation[edit]
Due to its high density, diiodomethane is employed in the separation of mineral samples by density. This application is particularly useful in geology and mineralogy for the identification and purification of mineral specimens.
NMR Spectroscopy[edit]
Diiodomethane is also used as an internal standard in NMR spectroscopy due to its distinct chemical shifts, which help in the accurate determination of molecular structures.
Safety[edit]
Diiodomethane is considered a hazardous substance and requires careful handling. It is toxic if ingested, inhaled, or absorbed through the skin. Safety precautions include the use of gloves, goggles, and adequate ventilation when handling the chemical. It is also a potential carcinogen and should be handled with utmost care.
Environmental Impact[edit]
The environmental impact of diiodomethane is a concern due to its toxicity and potential for bioaccumulation. Proper disposal methods should be followed to minimize its release into the environment.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
