Dequalinium
An article about the antiseptic and disinfectant compound Dequalinium
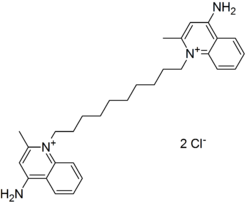
Dequalinium is a quaternary ammonium compound used primarily as an antiseptic and disinfectant. It is commonly found in the form of dequalinium chloride, which is used in various medical and dental applications due to its broad-spectrum antimicrobial properties.
Chemical Properties[edit]
Dequalinium is a cationic surfactant that belongs to the class of quaternary ammonium compounds. Its chemical structure allows it to interact with the cell membranes of bacteria, leading to cell lysis and death. This makes it effective against a wide range of gram-positive and gram-negative bacteria, as well as some fungi.
Medical Uses[edit]
Dequalinium is used in several medical applications, primarily for its antiseptic properties. It is often found in:
- Throat lozenges: Used to relieve symptoms of sore throat and pharyngitis.
- Vaginal tablets: Used to treat bacterial vaginosis and other vaginal infections.
- Mouthwashes and oral hygiene products: Helps in reducing oral bacteria and maintaining oral health.
Mechanism of Action[edit]
Dequalinium works by disrupting the cell membranes of microorganisms. The positively charged quaternary ammonium group interacts with the negatively charged components of the bacterial cell membrane, leading to increased permeability and eventual cell death. This mechanism is effective against a broad spectrum of microorganisms, making dequalinium a versatile antiseptic agent.
Safety and Side Effects[edit]
Dequalinium is generally well-tolerated when used as directed. However, some individuals may experience mild side effects such as:
- Irritation or burning sensation at the site of application
- Allergic reactions in sensitive individuals
It is important to use dequalinium products as recommended by healthcare professionals to minimize the risk of adverse effects.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian