Bromadiolone
Bromadiolone is a potent anticoagulant rodenticide that was first introduced in the 1970s. It is a vitamin K antagonist, which means it inhibits the action of vitamin K, leading to the depletion of clotting factors necessary for blood coagulation. This results in uncontrolled bleeding and eventually death in rodents. Bromadiolone is classified as a second-generation anticoagulant, which indicates it is more potent and has a longer half-life than first-generation anticoagulants, such as warfarin.
The use of bromadiolone is widespread in pest control to combat rodent populations in both urban and rural settings. It is available in various formulations, including baits that are placed in areas where rodent activity is observed. The effectiveness of bromadiolone, along with its delayed action (death occurs several days after ingestion), makes it a preferred choice in many rodent control programs. However, its potency and persistence raise significant concerns regarding environmental impact, particularly the risk of secondary poisoning to non-target species such as birds of prey and mammals that consume poisoned rodents.
Environmental protection agencies and regulatory bodies in many countries have guidelines and restrictions in place to mitigate the environmental risks associated with bromadiolone use. These may include restrictions on outdoor use, requirements for tamper-resistant bait stations, and mandates for users to have appropriate training or certification.
Despite its effectiveness in rodent control, the use of bromadiolone and other second-generation anticoagulants is a subject of ongoing debate. Research continues into their environmental impact, the development of resistance in rodent populations, and the search for alternative pest control methods that are both effective and have a lower environmental footprint.
See also[edit]
Bromadiolone[edit]
-
Skull and crossbones hazard symbol
-
Exclamation mark hazard symbol
-
Warning sign for rat poison
-
Bromadiolone paste
-
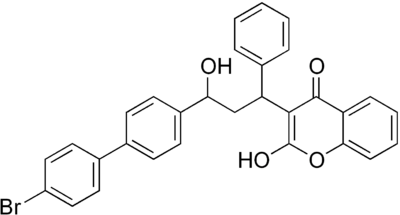
(R,S)-Bromadiolone structure
-
(S,R)-Bromadiolone structure
-
(R,R)-Bromadiolone structure
-
(S,S)-Bromadiolone structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian








