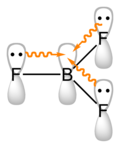
Boron trifluoride
Boron Trifluoride is a binary compound of boron and fluorine with the chemical formula BF3. This colorless gas is a valuable reagent in organic synthesis and has a pungent odor.
Chemical Properties[edit]
Boron Trifluoride is a planar molecule due to the arrangement of the three fluorine atoms around the central boron atom. The boron atom is sp2 hybridized, resulting in a trigonal planar geometry. The molecule is electron deficient with an empty p-orbital perpendicular to the plane of the molecule.
Physical Properties[edit]
Boron Trifluoride is a colorless, toxic gas at room temperature and pressure. It has a boiling point of -100.3°C and a melting point of -126.8°C. It is highly soluble in water and forms hydrofluoric acid and boric acid upon hydrolysis.
Production[edit]
Boron Trifluoride is primarily produced by the reaction of boron oxides with hydrogen fluoride. The reaction is highly exothermic and is carried out in a fluidized bed reactor.
Applications[edit]
Boron Trifluoride is widely used as a catalyst in organic synthesis for the preparation of various organic compounds. It is also used in the manufacture of high-performance fibers, in the petrochemical industry, and in the production of boron-containing materials.
Safety[edit]
Boron Trifluoride is a highly corrosive and toxic gas. It can cause severe burns and eye damage. It is also harmful if inhaled and can cause respiratory irritation.
See Also[edit]
```
Boron trifluoride[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian