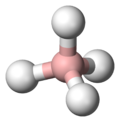
Borohydride
Borohydride refers to a class of chemical compounds that are characterized by the presence of a boron (B) atom bonded to four hydrogen (H) atoms, forming the borohydride anion (BH4−). These compounds are widely used in various chemical reactions, particularly as reducing agents in organic and inorganic chemistry due to their ability to donate hydride ions (H−).
Overview[edit]
Borohydrides are known for their versatility and efficiency in reducing a wide range of organic compounds, including aldehydes, ketones, and carboxylic acids, to their corresponding alcohols. They are also employed in the reduction of esters and amides to alcohols and amines, respectively. The most commonly used borohydride is sodium borohydride (NaBH4), but other derivatives such as lithium borohydride (LiBH4), potassium borohydride (KBH4), and calcium borohydride (Ca(BH4)2) are also significant.
Chemistry[edit]
The chemistry of borohydrides is centered around the borohydride ion (BH4−), which acts as a source of hydride ions. These ions are highly nucleophilic, making them effective in reducing electrophilic compounds. The reactivity and selectivity of borohydride reductions can be influenced by the choice of solvent, temperature, and the specific borohydride compound used.
Synthesis[edit]
Borohydrides can be synthesized through various methods, including the direct reaction of diborane (B2H6) with alkali metals or alkali metal hydrides. Another common method involves the reduction of borate salts with hydride donors such as lithium aluminium hydride (LiAlH4).
Applications[edit]
In addition to their widespread use in organic synthesis, borohydrides find applications in other areas such as:
- Fuel cells, where they serve as hydrogen storage materials.
- Pharmaceuticals, in the synthesis of various drugs.
- Material science, for the preparation of metal borides and other boron-containing compounds.
Safety and Handling[edit]
Borohydrides are generally stable under normal conditions but can be highly reactive with water, releasing hydrogen gas and potentially causing fire or explosion. Proper safety measures, including the use of inert atmospheres and dry solvents, are essential when handling these compounds.
Environmental Impact[edit]
The environmental impact of borohydrides is relatively low compared to other chemical reagents. However, the handling and disposal of these compounds should be conducted in a manner that minimizes exposure to the environment and complies with local regulations.
-
Borohydride
-
Zirconium borohydride
-
Beryllium borohydride complex
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian