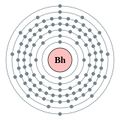
Bohrium
Bohrium is a synthetic chemical element with the symbol Bh and atomic number 107. It is a member of the transition metals and is part of the 7th period in the periodic table. Bohrium is named after the Danish physicist Niels Bohr, who made foundational contributions to understanding atomic structure and quantum theory.
Properties[edit]
Bohrium is a radioactive element and is not found naturally on Earth. It is produced artificially in a laboratory setting. Due to its position in the periodic table, it is expected to have properties similar to other group 7 elements, such as rhenium and manganese.
Physical Properties[edit]
- - Atomic Number: 107
- - Atomic Weight: [270] (most stable isotope)
- - Density: Unknown, but predicted to be around 37.1 g/cm³
- - Melting Point: Unknown
- - Boiling Point: Unknown
Chemical Properties[edit]
Bohrium is expected to exhibit chemical behavior similar to that of rhenium. It is predicted to form a stable +7 oxidation state, and possibly lower oxidation states as well. Due to its short half-life, detailed chemical studies have not been conducted.
Isotopes[edit]
Bohrium has several isotopes, all of which are radioactive. The most stable isotope is Bohrium-270, with a half-life of approximately 61 seconds. Other isotopes include Bohrium-267, Bohrium-271, and Bohrium-272, each with varying half-lives and decay modes.
Production[edit]
Bohrium is produced in particle accelerators through the fusion of lighter elements. The most common method involves bombarding Bismuth-209 with Chromium-54 ions:
\[ \text{\(^{209}_{83}Bi + \ ^{54}_{24}Cr \rightarrow \ ^{262}_{107}Bh + 1n\)} \]
This reaction produces Bohrium-262, which decays rapidly into lighter elements.
Applications[edit]
Due to its short half-life and the difficulty in producing it, bohrium has no practical applications outside of scientific research. It is primarily used in experiments to study the properties of superheavy elements and to test theoretical models of atomic structure.
History[edit]
Bohrium was first synthesized in 1981 by a team of scientists at the Gesellschaft für Schwerionenforschung (GSI) in Darmstadt, Germany. The team was led by Peter Armbruster and Gottfried Münzenberg. The element was named in honor of Niels Bohr, reflecting his contributions to the understanding of atomic structure.
See Also[edit]
-
Niels Bohr
-
Bohrium hassium meitnerium ceremony
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
- Budget GLP-1 shots
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian