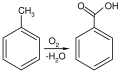
Benzoic acid
Benzoic Acid is a colorless crystalline solid and a simple aromatic carboxylic acid. It has the chemical formula C7H6O2. The name is derived from gum benzoin, which was for a long time its only known source. Benzoic acid occurs naturally in many plants and it serves as an intermediate in the biosynthesis of many secondary metabolites.
History[edit]
Benzoic acid was discovered in the sixteenth century. The dry distillation of gum benzoin was first described by Nostradamus (1556), and then by Alexius Pedemontanus (1560) and Blaise de Vigenère (1596).
Properties[edit]
Benzoic acid is a weak acid, stronger than the fatty acids, susceptible to oxidation, and reactive with bases to form salts. It is lipophilic and tends to accumulate in fatty tissues.
Production[edit]
Commercially, benzoic acid is produced by partial oxidation of toluene with oxygen. The process is catalyzed by cobalt or manganese naphthenates. The process uses cheap raw materials, proceeds in high yield, and is considered environmentally friendly.
Uses[edit]
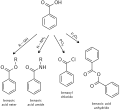
Benzoic acid is mainly consumed in the production of phenol by oxidative decarboxylation at 300−400 °C. The temperature required can be lowered to 200 °C by the addition of catalytic amounts of copper(II) salts. The phenol can be converted to cyclohexanol, which is a starting material for nylon synthesis.
Safety[edit]
Benzoic acid and its salts are generally recognized as safe (GRAS) by the Food and Drug Administration (FDA). They are used as food preservatives, with an E number of E210.
See also[edit]
Benzoic_acid[edit]
-
Pile of benzoic acid crystals
-
Benzoic acid chemical synthesis 1
-
Benzoic acid chemical reaction 1
-
Benzoic acid chemical reaction 2
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian