Atibeprone
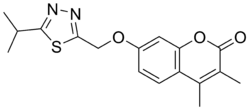
Atibeprone is a pharmacological drug that was developed in the late 20th century. It is classified as an antidepressant, specifically a non-selective monoamine reuptake inhibitor.
History[edit]
Atibeprone was first synthesized in the 1970s by the pharmaceutical company Upjohn. It was part of a series of compounds developed in an attempt to create a new class of antidepressants. However, despite showing promise in early clinical trials, Atibeprone was never marketed.
Pharmacology[edit]
Atibeprone acts by inhibiting the reuptake of monoamines, a group of neurotransmitters that includes serotonin, norepinephrine, and dopamine. By blocking the reuptake of these neurotransmitters, Atibeprone increases their concentration in the synaptic cleft, enhancing neurotransmission and leading to an improvement in mood.
Clinical Trials[edit]
In early clinical trials, Atibeprone showed promise as an antidepressant. It was found to be effective in reducing symptoms of depression and had a favorable side effect profile. However, further development of the drug was halted for unknown reasons, and it was never marketed.
Side Effects[edit]
The side effects of Atibeprone are similar to those of other non-selective monoamine reuptake inhibitors. These can include nausea, dizziness, insomnia, and anxiety. In rare cases, Atibeprone may also cause serotonin syndrome, a potentially life-threatening condition characterized by agitation, hallucinations, rapid heartbeat, and high body temperature.
Current Status[edit]
Despite its initial promise, Atibeprone is not currently available for use. The reasons for this are not entirely clear, but may relate to the drug's side effect profile, difficulties in manufacturing, or a lack of commercial interest.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
