Arsenic acid
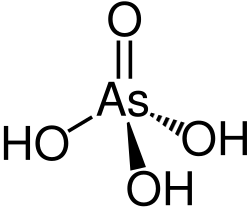
Arsenic acid is a chemical compound with the formula H₃AsO₄. It is a colorless, odorless, and highly toxic substance that is used in various industrial and agricultural applications. Arsenic acid is the arsenic analog of phosphoric acid and is a weak acid.
Chemical Properties[edit]
Arsenic acid is a triprotic acid, meaning it can donate three protons (H⁺ ions). The dissociation of arsenic acid in water occurs in three steps:
- H₃AsO₄ ⇌ H₂AsO₄⁻ + H⁺
- H₂AsO₄⁻ ⇌ HAsO₄²⁻ + H⁺
- HAsO₄²⁻ ⇌ AsO₄³⁻ + H⁺
The pKa values for these dissociations are approximately 2.2, 6.9, and 11.5, respectively.
Production[edit]
Arsenic acid can be produced by the oxidation of arsenic trioxide (As₂O₃) with concentrated nitric acid (HNO₃). The reaction is as follows: As₂O₃ + 2 HNO₃ + 2 H₂O → 2 H₃AsO₄
Uses[edit]
Arsenic acid is used in the manufacturing of wood preservatives, insecticides, and herbicides. It is also employed in the glass industry to remove bubbles from molten glass and in the semiconductor industry for the doping of silicon wafers.
Toxicity[edit]
Arsenic acid is highly toxic and poses significant health risks. Exposure to arsenic acid can lead to arsenic poisoning, which can cause symptoms such as nausea, vomiting, abdominal pain, and diarrhea. Chronic exposure can result in more severe health issues, including cancer, cardiovascular disease, and neuropathy.
Safety Measures[edit]
Due to its toxicity, handling arsenic acid requires strict safety measures. Personal protective equipment (PPE) such as gloves, goggles, and protective clothing should be worn. Proper ventilation and fume hoods should be used to minimize inhalation exposure. In case of contact with skin or eyes, immediate washing with plenty of water is necessary.
Environmental Impact[edit]
Arsenic acid can have detrimental effects on the environment. It can contaminate soil and water sources, leading to the bioaccumulation of arsenic in plants and animals. This can disrupt ecosystems and pose risks to human health through the food chain.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian