Antimony pentafluoride
Antimony pentafluoride is a chemical compound with the formula SbF5. It is one of the main fluorides of antimony, the others being SbF3 and SbF6−. This compound is a colorless, viscous liquid at room temperature, known for its strong Lewis acid properties and its ability to form complexes with almost all halide ions. Antimony pentafluoride plays a significant role in various chemical reactions, particularly in the field of organic chemistry and in the production of fluorinated compounds.
Properties[edit]
Antimony pentafluoride is characterized by its high reactivity and strong Lewis acidity. It is a powerful fluorinating agent, capable of converting many metal oxides to the corresponding metal fluorides and organic compounds to their fluorinated derivatives. The compound is highly corrosive to skin and mucous membranes and requires careful handling.
Synthesis[edit]
The synthesis of antimony pentafluoride involves the reaction of antimony trifluoride (SbF3) with fluorine gas. The process requires controlled conditions due to the high reactivity of the reactants and the corrosive nature of the product.
Applications[edit]
Antimony pentafluoride is used in various applications due to its strong Lewis acid properties. It is utilized in the synthesis of organofluorine compounds, as a catalyst in the alkylation of hydrocarbons, and in the production of superacids when combined with hydrogen fluoride. Its ability to form stable complexes with fluoride ions makes it useful in nuclear reactor fuel processing and in the study of coordination chemistry.
Safety[edit]
Due to its corrosive nature and high reactivity, antimony pentafluoride requires careful handling. Appropriate safety measures, including the use of personal protective equipment (PPE), are essential when working with this compound to prevent exposure and chemical burns.
Antimony pentafluoride gallery[edit]
-
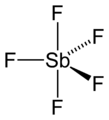
Antimony pentafluoride 2D structure
-
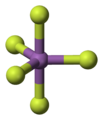
Antimony pentafluoride monomer 3D balls
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian