4-Aminopyridine
Chemical compound used in medicine and research
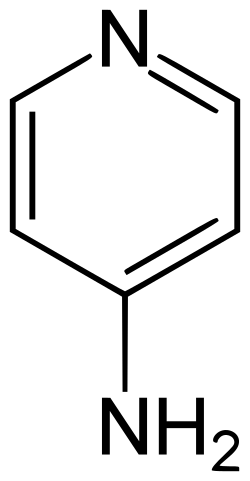
4-Aminopyridine (also known as fampridine) is a chemical compound with the formula C5H6N2. It is a derivative of pyridine and is used in various medical and research applications. This compound is known for its ability to block potassium channels, which has implications in the treatment of certain neurological disorders.
Chemical Properties[edit]
4-Aminopyridine is a white crystalline powder that is soluble in water and organic solvents. It has a melting point of approximately 158-160 °C. The compound is stable under normal conditions but should be stored in a cool, dry place to prevent degradation.
Medical Uses[edit]
4-Aminopyridine is primarily used in the treatment of multiple sclerosis (MS). It functions by improving nerve conduction in demyelinated nerves, which can help alleviate symptoms such as muscle weakness and fatigue. The drug is marketed under the name Ampyra in the United States and Fampyra in Europe.
Mechanism of Action[edit]
The primary mechanism of action of 4-aminopyridine is the blockade of voltage-gated potassium channels. By inhibiting these channels, the compound prolongs the action potential in neurons, thereby enhancing synaptic transmission and improving neurological function.
Research Applications[edit]
In addition to its medical uses, 4-aminopyridine is employed in research settings to study the function of potassium channels and their role in various physiological processes. It is also used in the development of new therapeutic agents targeting neurological disorders.
Safety and Side Effects[edit]
While 4-aminopyridine is effective in treating certain conditions, it can cause side effects such as dizziness, insomnia, and nausea. In some cases, it may lead to more severe reactions like seizures, especially at higher doses. Patients using this medication should be monitored closely by healthcare professionals.
Synthesis[edit]
The synthesis of 4-aminopyridine involves the amination of pyridine derivatives. Various methods have been developed to achieve this transformation, often involving the use of catalysts and specific reaction conditions to optimize yield and purity.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian