Aminoethylpiperazine
Aminoethylpiperazine is a derivative of piperazine. This chemical compound is commonly used in the manufacture of resins, epoxy curing agents, and surfactants.
Chemical Structure[edit]
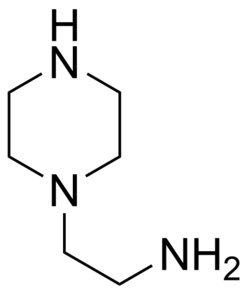
Aminoethylpiperazine is a secondary amine with the chemical formula C6H15N3. It is a clear, colorless liquid that is soluble in water and most organic solvents. The molecule consists of a piperazine ring, with an amino group (NH2) attached to one of the carbon atoms.
Uses[edit]
Aminoethylpiperazine is primarily used in the production of epoxy curing agents. These are substances that are added to epoxy resins to cause them to harden. It is also used as a building block in the synthesis of surfactants, which are compounds that lower the surface tension of a liquid, allowing it to spread more easily.
In addition, Aminoethylpiperazine can be used as a corrosion inhibitor, preventing damage to metals by halting the chemical reaction that causes corrosion. It is also used in the production of polyamide resins and as a catalyst in the production of flexible urethane foams.
Safety[edit]
Aminoethylpiperazine is a corrosive substance, and can cause burns to the skin and eyes. It is also harmful if inhaled or swallowed. Therefore, it is important to handle this chemical with care, using appropriate personal protective equipment and following safety procedures.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
