Acotiamide
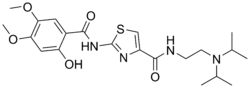
Acotiamide is a drug used primarily for the treatment of functional dyspepsia. It is developed by Zeria Pharmaceutical Co. Ltd in Japan and marketed under the brand name Acofide. Acotiamide works by enhancing gastric motility and is classified as a prokinetic agent.
Mechanism of Action[edit]
Acotiamide works by inhibiting the enzymes acetylcholinesterase (AChE) and muscarinic M1 and M3 receptors. This inhibition leads to an increase in the concentration of acetylcholine in the synaptic cleft, which in turn enhances gastric motility. Acotiamide does not have any effect on gastric acid secretion.
Clinical Use[edit]
Acotiamide is used for the treatment of functional dyspepsia, a condition characterized by chronic or recurrent pain or discomfort centered in the upper abdomen. Symptoms of functional dyspepsia include postprandial fullness, early satiety, and epigastric pain or burning. Acotiamide has been shown to improve these symptoms and enhance gastric emptying.
Side Effects[edit]
The most common side effects of acotiamide include constipation, dizziness, and rash. These side effects are generally mild and transient. Acotiamide is contraindicated in patients with known hypersensitivity to the drug.
Pharmacokinetics[edit]
Acotiamide is rapidly absorbed after oral administration, with peak plasma concentrations reached within 1 to 2 hours. The drug is extensively metabolized in the liver, primarily by the enzyme CYP3A4. The elimination half-life of acotiamide is approximately 4 to 5 hours.
Regulatory Status[edit]
Acotiamide was approved by the Japanese Ministry of Health, Labour and Welfare in 2012 for the treatment of functional dyspepsia. As of 2021, it is not approved for use in the United States or Europe.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
