Acid dissociation constant
Acid Dissociation Constant[edit]
The acid dissociation constant, denoted as Ka, is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction known as dissociation in the context of acid-base reactions. The equilibrium is represented as:
- HA _ H+ + A-
where HA is a generic acid that dissociates into a proton (H+) and its conjugate base (A-). The larger the value of Ka, the more the acid dissociates, and the stronger the acid.
Definition and Expression[edit]
The acid dissociation constant is defined by the equation:
- Ka = \( \frac{[\text{H}^+][\text{A}^-]}{[\text{HA}]} \)
where [H+], [A-], and [HA] are the molar concentrations of the hydrogen ion, the conjugate base, and the undissociated acid, respectively.
pKa[edit]
The pKa is the negative logarithm (base 10) of the acid dissociation constant:
- pKa = -log10Ka
A lower pKa value indicates a stronger acid, which means it more fully dissociates in solution.
Factors Affecting Acid Strength[edit]
Several factors influence the strength of an acid, including:
- Electronegativity: More electronegative atoms can stabilize the negative charge on the conjugate base, increasing acid strength.
- Resonance: Delocalization of charge through resonance can stabilize the conjugate base.
- Inductive Effect: Electron-withdrawing groups can stabilize the conjugate base through the inductive effect.
- Hybridization: The s-character of the hybrid orbitals can affect acidity; more s-character can lead to stronger acids.
Examples of Acid Dissociation Constants[edit]
Acetic Acid[edit]
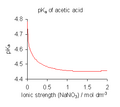
Acetic acid (CH3COOH) is a weak acid with a pKa of approximately 4.76. It partially dissociates in water to form acetate ions (CH3COO-) and hydrogen ions (H+).
Phosphoric Acid[edit]

Phosphoric acid (H3PO4) is a triprotic acid with three dissociation constants, corresponding to the loss of each proton.
Citric Acid[edit]
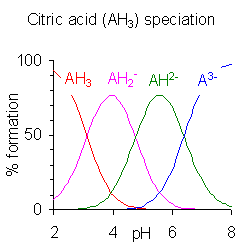
Citric acid (C6H8O7) is a weak organic acid with three carboxyl groups, each with its own dissociation constant.
Related Concepts[edit]
Related Pages[edit]
Gallery[edit]
-
pKa of acetic acid
-
Weak acid speciation
-
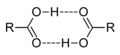
Carboxylic acid dimers
-
Acetic acid pKa in dioxane-water
-
Chloroacetic acid pKa
-
Fumaric acid structure
-
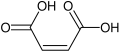
Maleic acid structure
-
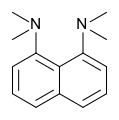
Proton sponge
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
-
Acid dissociation constant
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian