Absorption spectroscopy
Absorption spectroscopy
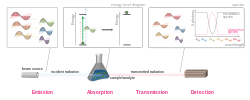
Absorption spectroscopy is a technique used in analytical chemistry to determine the presence and concentration of a substance in a sample by measuring the amount of light absorbed by the sample. This method is based on the principle that atoms and molecules absorb light at specific wavelengths, which correspond to the energy differences between their electronic states.
Principles of Absorption Spectroscopy[edit]
Absorption spectroscopy involves the interaction of light with matter. When light passes through a sample, certain wavelengths are absorbed by the sample's atoms or molecules. The amount of light absorbed at each wavelength is measured and used to create an absorption spectrum. This spectrum can be used to identify the substance and determine its concentration.
The basic components of an absorption spectrometer include a light source, a sample holder, a monochromator to select specific wavelengths of light, and a detector to measure the intensity of transmitted light.
Types of Absorption Spectroscopy[edit]
There are several types of absorption spectroscopy, each suited to different applications:
- UV-Vis spectroscopy: Measures absorption in the ultraviolet and visible regions of the electromagnetic spectrum. It is commonly used for quantitative analysis of solutions.
- IR spectroscopy: Measures absorption in the infrared region, providing information about molecular vibrations and structure.
- Atomic absorption spectroscopy (AAS): Used to analyze metal ions in solutions by measuring the absorption of light by free atoms.
Applications[edit]
Absorption spectroscopy is widely used in various fields, including:
- Environmental science: Monitoring pollutants in air and water.
- Pharmaceutical industry: Quality control and drug development.
- Astronomy: Identifying elements in stars and planets.

Astronomical Applications[edit]
In astronomy, absorption spectroscopy is used to study the composition of stars, planets, and other celestial bodies. By analyzing the absorption lines in the spectra of stars, astronomers can determine the elements present in their atmospheres.
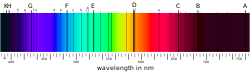
Spectral Lines[edit]
Absorption lines, such as the Fraunhofer lines in the solar spectrum, are dark lines that appear in the spectrum when light is absorbed by atoms or molecules. These lines are characteristic of specific elements and can be used to identify them.
Gallery[edit]
-
Emission spectrum of iron, showing lines that correspond to absorption lines.
-
Identification of ices in the solar system using absorption spectroscopy.
-
Cumulative absorption spectrum from the Hubble Space Telescope.
Related Pages[edit]
Absorption_spectroscopy[edit]
-
Overview of spectroscopy techniques
-
Sodium in the atmosphere of exoplanet HD 209458
-
Fraunhofer lines in the solar spectrum
-
Emission spectrum of iron (Fe)
-
Identification of ices in the Solar System
-
Cumulative absorption spectrum from the Hubble Telescope

