5α-Dihydrolevonorgestrel
A synthetic progestogen
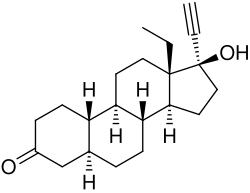
5α-Dihydrolevonorgestrel is a synthetic progestogen and a metabolite of the hormonal contraceptive levonorgestrel. It is part of the class of steroids known as 19-nortestosterone derivatives. This compound is notable for its role in the pharmacokinetics and pharmacodynamics of levonorgestrel, contributing to its biological activity in the body.
Chemical Structure and Properties[edit]
5α-Dihydrolevonorgestrel is a steroid with a chemical structure that is closely related to that of levonorgestrel. It is characterized by the reduction of the double bond in the A-ring of the steroid nucleus, resulting in a fully saturated A-ring. This structural modification is responsible for its distinct pharmacological properties compared to its parent compound.
Pharmacology[edit]
As a metabolite of levonorgestrel, 5α-Dihydrolevonorgestrel plays a role in the mechanism of action of this widely used contraceptive. Levonorgestrel is metabolized in the liver, where it undergoes reduction to form 5α-Dihydrolevonorgestrel. This metabolite retains progestogenic activity, contributing to the overall contraceptive effect by inhibiting ovulation, altering the endometrium, and thickening cervical mucus.
Clinical Significance[edit]
The presence of 5α-Dihydrolevonorgestrel in the body is an important factor in the effectiveness of levonorgestrel-based contraceptives. Understanding the metabolism and activity of this compound helps in optimizing the dosing and administration of levonorgestrel to achieve desired contraceptive outcomes. Additionally, the study of its pharmacokinetics can provide insights into individual variations in response to hormonal contraceptives.
Related Compounds[edit]
5α-Dihydrolevonorgestrel is related to other 19-nortestosterone derivatives, which include a variety of synthetic progestogens used in hormonal contraception and hormone replacement therapy. These compounds share a common structural framework but differ in their specific chemical modifications, which influence their pharmacological profiles.
Research and Development[edit]
Research into 5α-Dihydrolevonorgestrel and its parent compound, levonorgestrel, continues to be an area of interest in the field of reproductive health. Studies focus on improving the efficacy and safety of contraceptive methods, understanding the metabolic pathways involved, and exploring potential new applications for these compounds in other therapeutic areas.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian