3-Methyl-2-butanol
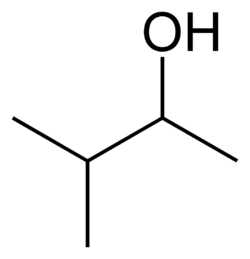
3-Methyl-2-butanol, also known as isopentyl alcohol, is an organic compound with the formula C5H12O. It is one of several isomers of amyl alcohol. This colorless liquid is a common solvent and an intermediate in the manufacture of other chemicals.
Properties[edit]
3-Methyl-2-butanol is a secondary alcohol that is flammable and has a characteristic strong, fusel oil odor. It is less dense than water and soluble in water, which allows it to be used in various applications in the chemical industry. Its boiling point is relatively high compared to other alcohols of similar molecular weight, which makes it useful in applications that require a solvent with a moderate evaporation rate.
Production[edit]
The production of 3-Methyl-2-butanol typically involves the hydroformylation of isobutene to form isovaleraldehyde, followed by hydrogenation to produce the alcohol. This process is part of the larger oxo alcohol synthesis, which is a key method for producing alcohols from alkenes.
Applications[edit]
3-Methyl-2-butanol is used in a variety of applications due to its solvent properties. It is utilized in the manufacture of paints, varnishes, and plastics, as well as in the formulation of hydraulic fluids. Additionally, it serves as a starting material in the synthesis of flavors and fragrances, pharmaceuticals, and other fine chemicals.
Safety[edit]
Like many organic solvents, 3-Methyl-2-butanol poses health risks if inhaled, ingested, or comes into contact with the skin. It can cause irritation to the eyes, skin, and respiratory system. Proper handling and storage procedures must be followed to minimize exposure and reduce the risk of fire and health hazards.
Environmental Impact[edit]
The environmental impact of 3-Methyl-2-butanol includes its potential to contribute to air and water pollution if not properly managed. It is biodegradable under aerobic conditions, but its release into the environment should be avoided to prevent potential harm to aquatic life and the broader ecosystem.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
