2-Undecanone
2-Undecanone[edit]
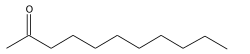
2-Undecanone, also known as methyl nonyl ketone, is an organic compound with the chemical formula C11H22O. It is a ketone that is commonly used in the fragrance and flavor industries due to its distinctive odor. 2-Undecanone is also known for its role as a natural insect repellent.
Chemical Properties[edit]
2-Undecanone is a ketone, characterized by the presence of a carbonyl group (C=O) bonded to two hydrocarbon groups. In the case of 2-Undecanone, the carbonyl group is bonded to a methyl group and a nonyl group, making it a methyl ketone. It is a colorless to pale yellow liquid at room temperature.
Occurrence[edit]
2-Undecanone occurs naturally in several plants, including wild tomato species, where it serves as a defense mechanism against herbivores and insects. It is also found in the essential oils of some plants and is used in the formulation of perfumes and flavors.
Uses[edit]
Insect Repellent[edit]
2-Undecanone is used as a natural insect repellent. It is effective against a variety of insects, including mosquitoes, ticks, and flies. Its use as a repellent is particularly valued in organic farming and gardening, where synthetic chemicals are avoided.
Fragrance and Flavor[edit]
In the fragrance industry, 2-Undecanone is used to impart a fruity or floral scent to perfumes and other scented products. In the flavor industry, it is used to add a fruity note to food products.
Synthesis[edit]
2-Undecanone can be synthesized through various chemical processes, including the oxidation of undecanol or the Friedel-Crafts acylation of nonane with acetyl chloride.
Safety[edit]
While 2-Undecanone is generally considered safe for use in consumer products, it should be handled with care in its pure form. It can cause irritation to the skin and eyes upon contact.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian