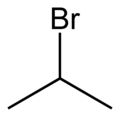
2-Bromopropane
2-Bromopropane[edit]


2-Bromopropane, also known as isopropyl bromide, is an organobromine compound with the chemical formula C_H_Br. It is a colorless liquid that is used in various industrial applications, including as a solvent and an intermediate in organic synthesis.
Structure and Properties[edit]
2-Bromopropane is a haloalkane with a molecular structure consisting of a three-carbon alkane chain with a bromine atom attached to the second carbon. This structure gives it the IUPAC name 2-bromopropane. The presence of the bromine atom makes it a polar molecule, which influences its physical and chemical properties.
Physical Properties[edit]
2-Bromopropane is a colorless liquid at room temperature. It has a boiling point of approximately 59 °C and a density of about 1.31 g/cm_. It is slightly soluble in water but miscible with most organic solvents.
Chemical Properties[edit]
As a haloalkane, 2-bromopropane is reactive in nucleophilic substitution reactions. It can undergo SN1 and SN2 reactions, depending on the conditions and the nucleophile involved. It is also susceptible to elimination reactions, which can lead to the formation of alkenes.
Synthesis[edit]
2-Bromopropane can be synthesized through the halogenation of propane using bromine in the presence of light or heat. Alternatively, it can be prepared by the reaction of isopropanol with hydrogen bromide in the presence of a strong acid catalyst.
Uses[edit]
2-Bromopropane is used in various industrial applications:
- As a solvent in the manufacturing of pharmaceuticals and agrochemicals.
- As an intermediate in the synthesis of other organic compounds.
- In the production of flame retardants and plasticizers.
Health and Safety[edit]
Exposure to 2-bromopropane can pose health risks. It is important to handle it with care, using appropriate personal protective equipment and following safety guidelines. Prolonged exposure can lead to respiratory and dermatological issues.
Related Pages[edit]
Gallery[edit]
-
2D skeletal structure of 2-Bromopropane
-
2D flat structure of 2-Bromopropane
-
2-Bromopropane skeletal structure
-
2-Bromopropane flat structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian