2-Aminopurine
2-Aminopurine[edit]
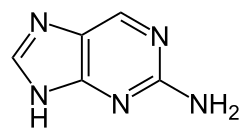
2-Aminopurine is a purine analog that is used in molecular biology and genetics research. It is a derivative of adenine, one of the four nucleobases in the nucleic acid of DNA and RNA. 2-Aminopurine is known for its ability to act as a fluorescent probe and its role as a mutagen.
Chemical Properties[edit]
2-Aminopurine is a structural analog of adenine, differing by the presence of an amino group at the 2-position of the purine ring. This modification allows it to pair with both thymine and cytosine, which can lead to mutations during DNA replication.
Applications in Research[edit]
2-Aminopurine is widely used in research due to its fluorescent properties. It can be incorporated into nucleic acids and used to study DNA-protein interactions, nucleic acid dynamics, and enzyme mechanisms. Its fluorescence is sensitive to the local environment, making it a valuable tool for probing the structure and dynamics of nucleic acids.
Mutagenic Effects[edit]
As a mutagen, 2-Aminopurine can induce base-pair substitutions by mispairing during DNA replication. This property is utilized in genetic studies to induce mutations and study their effects on gene function and protein expression.
Safety and Handling[edit]
While 2-Aminopurine is a useful research tool, it is important to handle it with care due to its mutagenic properties. Proper laboratory safety protocols should be followed to minimize exposure and prevent contamination.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian