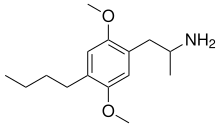
2,5-Dimethoxy-4-butylamphetamine
Chemical compound
| 2,5-Dimethoxy-4-butylamphetamine | |
|---|---|
|
2,5-Dimethoxy-4-butylamphetamine
| |
| Chemical nomenclature | |
| IUPAC name | 1-(4-butyl-2,5-dimethoxyphenyl)propan-2-amine
|
2,5-Dimethoxy-4-butylamphetamine (DOBU) is a psychedelic phenethylamine of the substituted amphetamines class. It is a chemical compound that has been studied for its psychoactive effects and potential use in psychopharmacology.
Chemical Structure[edit]
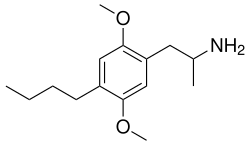
DOBU is structurally related to other psychedelic amphetamines such as 2,5-Dimethoxy-4-bromoamphetamine (DOB) and 2,5-Dimethoxy-4-iodoamphetamine (DOI). The chemical structure of DOBU includes a phenethylamine backbone with methoxy groups at the 2 and 5 positions and a butyl group at the 4 position of the aromatic ring.
Pharmacology[edit]
DOBU acts as a serotonin receptor agonist, primarily targeting the 5-HT2A receptor, which is believed to be responsible for its psychedelic effects. The interaction with serotonin receptors is similar to that of other compounds in the 2C family and DOx family, which are known for their hallucinogenic properties.
Effects[edit]
The effects of DOBU are not well-documented in scientific literature, but it is presumed to produce effects similar to other psychedelic amphetamines. These effects may include altered perception, mood changes, and visual hallucinations. The duration and intensity of these effects can vary based on dosage and individual sensitivity.
Synthesis[edit]
The synthesis of DOBU involves the alkylation of 2,5-dimethoxyphenethylamine with a butyl halide. This process is similar to the synthesis of other substituted amphetamines, where the choice of alkylating agent determines the specific substituent on the aromatic ring.
Legal Status[edit]
The legal status of DOBU varies by country. In some jurisdictions, it may be classified as a controlled substance due to its structural similarity to other regulated psychedelic compounds. Researchers must be aware of local regulations when studying or handling this compound.
Related Compounds[edit]
DOBU is part of a larger family of psychedelic amphetamines, including:
- 2,5-Dimethoxy-4-bromoamphetamine (DOB)
- 2,5-Dimethoxy-4-iodoamphetamine (DOI)
- 2,5-Dimethoxy-4-methylamphetamine (DOM)
These compounds share a common phenethylamine structure with variations in the substituents on the aromatic ring, which influence their pharmacological properties and effects.
See Also[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian