18F-EF5
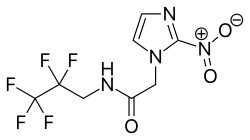
18F-EF5 is a radiopharmaceutical compound used in positron emission tomography (PET) imaging to detect hypoxia in tumor tissues. It is a fluorine-18 labeled derivative of the nitroimidazole compound EF5, which selectively binds to hypoxic cells.
Chemical Structure[edit]
18F-EF5 is a fluorinated analog of EF5, where the radioactive isotope fluorine-18 is incorporated into the molecular structure. The chemical structure allows it to penetrate cells and bind to intracellular components under low oxygen conditions.
Mechanism of Action[edit]
The primary mechanism of action for 18F-EF5 involves its selective binding to hypoxic cells. Hypoxia, a condition characterized by low oxygen levels, is a common feature of many solid tumors. Under hypoxic conditions, 18F-EF5 undergoes bioreduction and forms covalent bonds with intracellular macromolecules, allowing for the visualization of hypoxic regions within tumors using PET imaging.
Clinical Applications[edit]
18F-EF5 is primarily used in the field of oncology to assess tumor hypoxia. Hypoxia is a significant factor in tumor progression, resistance to therapy, and overall prognosis. By identifying hypoxic regions within tumors, clinicians can better tailor treatment strategies, such as radiotherapy and chemotherapy, to improve patient outcomes.
Synthesis[edit]
The synthesis of 18F-EF5 involves the incorporation of fluorine-18 into the EF5 molecule. This process typically requires a cyclotron to produce the fluorine-18 isotope, followed by a series of chemical reactions to attach the isotope to the EF5 compound. The final product is then purified and prepared for use in PET imaging.
Advantages[edit]
One of the main advantages of using 18F-EF5 in PET imaging is its high specificity for hypoxic cells. This allows for precise imaging of hypoxic regions within tumors, which can be critical for effective treatment planning. Additionally, the use of fluorine-18, with its relatively short half-life of approximately 110 minutes, minimizes radiation exposure to patients.
Limitations[edit]
Despite its advantages, 18F-EF5 has some limitations. The synthesis process is complex and requires specialized equipment and expertise. Additionally, the short half-life of fluorine-18 necessitates rapid synthesis and administration, which can be logistically challenging.
Future Directions[edit]
Research is ongoing to improve the synthesis and application of 18F-EF5. Advances in radiochemistry and imaging technology may enhance the utility of this compound in clinical practice. Additionally, studies are being conducted to explore the use of 18F-EF5 in other hypoxia-related conditions beyond oncology.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian