(R)-MDMA
(R)-MDMA[edit]
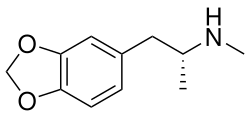
(R)-MDMA is one of the two enantiomers of the psychoactive drug MDMA, commonly known as "ecstasy". The compound is a chiral molecule, meaning it has a non-superimposable mirror image, and (R)-MDMA is the right-handed enantiomer. The other enantiomer is (S)-MDMA, and the two have different pharmacological effects.
Chemical Structure and Properties[edit]
(R)-MDMA, or (R)-3,4-methylenedioxy-N-methylamphetamine, is a synthetic compound with the chemical formula C11H15NO2. It is characterized by the presence of a methylenedioxy group attached to the phenyl ring and a methyl group attached to the nitrogen atom. The stereochemistry of (R)-MDMA is defined by the configuration of the chiral center at the alpha carbon of the side chain.
Pharmacology[edit]
The pharmacological effects of (R)-MDMA differ from those of its enantiomer, (S)-MDMA. While both enantiomers act as serotonin and dopamine releasers, (R)-MDMA is generally considered to be less potent in terms of its psychoactive effects. Studies suggest that (R)-MDMA has a lower affinity for the serotonin transporter (SERT) compared to (S)-MDMA, which may account for its reduced psychoactivity.
Effects and Uses[edit]
The effects of (R)-MDMA are less well-studied than those of the racemic mixture or (S)-MDMA alone. However, it is known that (R)-MDMA can produce mild stimulant effects, including increased energy and alertness. Unlike (S)-MDMA, which is associated with empathogenic effects, (R)-MDMA is less likely to produce feelings of emotional closeness or euphoria.
In research settings, (R)-MDMA is sometimes used to study the stereochemistry of MDMA and its effects on the brain. It is not commonly used recreationally due to its reduced psychoactive properties compared to the racemic mixture.
Synthesis[edit]
The synthesis of (R)-MDMA involves the resolution of the racemic mixture of MDMA or the use of chiral starting materials to produce the desired enantiomer. Various methods have been developed to achieve this, including the use of chiral catalysts or chiral auxiliaries in the synthetic process.
Legal Status[edit]
Like MDMA, (R)-MDMA is classified as a controlled substance in many countries. Its legal status is typically the same as that of the racemic mixture, as laws often do not distinguish between enantiomers of controlled substances.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian