Biginelli reaction
Biginelli Reaction
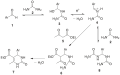
The Biginelli reaction is a significant chemical reaction that allows for the synthesis of dihydropyrimidinones (DHPMs), which are a class of heterocyclic compounds. This reaction was first reported in 1893 by Pietro Biginelli, an Italian chemist, hence its name. The Biginelli reaction is a multicomponent reaction (MCR), meaning it involves the simultaneous reaction of three or more starting materials to form a product, where the core structure is a 3,4-dihydropyrimidin-2(1H)-one. The typical substrates for this reaction include an aldehyde, a β-keto ester (or a β-diketone), and urea or thiourea.
Reaction Mechanism[edit]
The mechanism of the Biginelli reaction involves several steps, starting with the formation of an enamine from the β-keto ester and the aldehyde. This enamine then reacts with urea or thiourea to form an intermediate, which undergoes cyclization to produce the dihydropyrimidinone core structure. The reaction is typically acid-catalyzed, with various acids being used to promote the reaction, including both organic and inorganic acids.
Variations and Developments[edit]
Over the years, several modifications and improvements have been made to the original Biginelli reaction protocol to increase its efficiency, yield, and scope. These include the use of different catalysts, such as Lewis acids, Brønsted acids, and even heterogeneous catalysts. Additionally, microwave-assisted and solvent-free conditions have been explored to enhance the reaction rates and environmental friendliness of the process.
Applications[edit]
Dihydropyrimidinones, the products of the Biginelli reaction, exhibit a wide range of biological activities, including antihypertensive, antiviral, and anticancer properties. This makes the Biginelli reaction an important tool in medicinal chemistry for the synthesis of new pharmaceuticals. Furthermore, the reaction is used in the synthesis of calcium channel blockers, a class of medications used to treat high blood pressure.
See Also[edit]
External Links[edit]
Biginelli reaction gallery[edit]
-
Biginelli Reaction Scheme
-
Biginelli Reaction Mechanism
-
Biginelli reaction mechanism
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian