Lead(II) oxide
Lead(II) oxide, also known as lead monoxide or litharge, is a chemical compound with the formula PbO. It is a yellowish or reddish crystalline solid that is commonly used in various industrial applications. In this article, we will explore the properties, uses, and safety considerations of Lead(II) oxide.
Properties[edit]
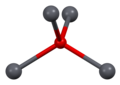
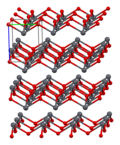
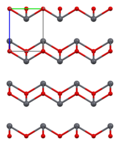
Lead(II) oxide has a molecular weight of 223.2 g/mol and a melting point of 888°C. It is insoluble in water but soluble in acids, forming lead salts. The compound exists in two polymorphs: litharge, which has a tetragonal crystal structure, and massicot, which has an orthorhombic structure.
Uses[edit]
Lead(II) oxide has a wide range of applications across different industries. One of its primary uses is in the production of lead-acid batteries, where it serves as a component of the positive electrode (anode). The compound helps to convert lead sulfate into lead dioxide during the charging process, allowing the battery to store and release electrical energy efficiently.
Another significant application of Lead(II) oxide is in the manufacturing of glass and ceramics. It acts as a flux, reducing the melting point of the materials and improving their workability. Additionally, the compound is used as a pigment in the production of yellow and red paints, as well as in the coloring of glassware.
Lead(II) oxide also finds use in the field of electronics. It is employed in the production of semiconductors, particularly in the fabrication of certain types of diodes and transistors. The compound's electrical properties make it suitable for these applications.
Safety Considerations[edit]
It is important to note that Lead(II) oxide is toxic and poses health risks if not handled properly. Inhalation or ingestion of the compound can lead to lead poisoning, which can cause severe health issues, especially in children and pregnant women. Therefore, it is crucial to follow safety guidelines and use appropriate protective equipment when working with or around Lead(II) oxide.
See Also[edit]
References[edit]
<references />
Lead(II) oxide[edit]
-
Lead(II) oxide - Pb coordination
-
Lead(II) oxide - O coordination
-
Lead(II) oxide - Unit cell
-
Lead(II) oxide - 3x3x3 structure
-
Lead(II) oxide - 3x3x3 structure (a-axis)
-
Lead(II) oxide - 3x3x3 structure (c-axis)
-
Lead(II) oxide
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian