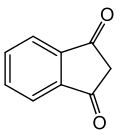
1,3-Indandione
Chemical compound
1,3-Indandione[edit]

1,3-Indandione is an organic compound with the molecular formula C9H6O2. It is a derivative of indan, a bicyclic compound, and is characterized by a diketone functional group. This compound is of interest in various chemical reactions and has applications in the synthesis of other chemical compounds.
Chemical Properties[edit]
1,3-Indandione is a yellow crystalline solid at room temperature. It is known for its ability to undergo various chemical reactions, including decarboxylation, reduction, and bromination. The compound is soluble in organic solvents and exhibits keto-enol tautomerism.
Synthesis[edit]
1,3-Indandione can be synthesized through several methods. One common method involves the decarboxylation of indan-1,3-dicarboxylic acid. The reaction is typically carried out under heat, resulting in the loss of carbon dioxide and the formation of 1,3-Indandione.

Reactions[edit]
1,3-Indandione participates in a variety of chemical reactions:
Reduction[edit]
The compound can be partially or fully reduced to form different products. Partial reduction leads to the formation of 1,3-indanediol, while complete reduction results in the formation of indane.

Bromination[edit]
Bromination of 1,3-Indandione involves the addition of bromine to the compound, resulting in the formation of brominated derivatives. This reaction is useful in the synthesis of more complex molecules.

Bindone Formation[edit]
1,3-Indandione can be used in the synthesis of bindone, a compound of interest in organic chemistry. The reaction involves the formation of a new carbon-carbon bond, expanding the indandione ring system.

Applications[edit]
1,3-Indandione is used in the synthesis of various pharmaceuticals and as an intermediate in organic synthesis. Its ability to undergo multiple chemical transformations makes it a valuable compound in the field of medicinal chemistry.
Related Pages[edit]
Gallery[edit]
-
3D model of 1,3-Indandione
-
Synthesis involving 1,3-Indandione
-
Complete reduction of 1,3-Indandione
1,3-Indandione[edit]
-
1,3-Indandione
-
1,3-Indandione
-
Indandione Decarboxylation
-
NaECI Preparation
-
Bindone Formation
-
Bromination of 1,3-Indanediol
-
Partial Reduction of 1,3-Indanedione
-
Reduction of 1,3-Indanedione
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian