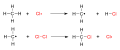Free-radical halogenation
Free-radical halogenation is a chemical reaction in which a halogen atom is introduced into an alkane or alkene by means of a free-radical mechanism. This process is a fundamental method for the organic synthesis of halogenated compounds and plays a critical role in the field of organic chemistry. The reaction is characterized by the substitution of a hydrogen atom in the hydrocarbon with a halogen atom (Fluorine, Chlorine, Bromine, or Iodine), initiated by the generation of free radicals.
Mechanism[edit]
The free-radical halogenation reaction proceeds through three main steps: initiation, propagation, and termination.
Initiation[edit]
The initiation step involves the homolytic cleavage of the halogen molecule (X_2) into two halogen radicals (X•) under the influence of heat, light, or a radical initiator. This process generates the reactive species that drives the reaction forward.
- \(\text{X}_2 \rightarrow 2 \text{X}•\)
Propagation[edit]
During the propagation step, a halogen radical abstracts a hydrogen atom from the hydrocarbon, forming a hydrocarbon radical and hydrohalic acid. The newly formed hydrocarbon radical then reacts with another molecule of the halogen to form the halogenated product and regenerate the halogen radical, perpetuating the reaction cycle.
- \(\text{X}• + \text{R-H} \rightarrow \text{R}• + \text{H-X}\)
- \(\text{R}• + \text{X}_2 \rightarrow \text{R-X} + \text{X}•\)
Termination[edit]
Termination occurs when two radicals combine to form a stable molecule, effectively removing them from the reaction mixture and ending the reaction. This can happen in various ways, such as two halogen radicals forming a dihalogen molecule or a halogen radical combining with a hydrocarbon radical.
- \(\text{X}• + \text{X}• \rightarrow \text{X}_2\)
- \(\text{R}• + \text{X}• \rightarrow \text{R-X}\)
Selectivity[edit]
The selectivity of free-radical halogenation is influenced by the type of halogen and the structure of the hydrocarbon. For instance, the reactivity of halogens decreases in the order of Fluorine > Chlorine > Bromine > Iodine, which is inversely related to the selectivity. Fluorine reacts explosively with most organic compounds, while iodine is relatively unreactive. Chlorine and bromine are commonly used for selective halogenation, with bromine being more selective due to its lower reactivity.
Applications[edit]
Free-radical halogenation is widely used in the synthesis of halogenated organic compounds, which serve as intermediates in the production of pharmaceuticals, agrochemicals, and polymers. It is also employed in the modification of hydrocarbons to improve their reactivity or physical properties.
Safety and Environmental Considerations[edit]
The use of halogens, particularly chlorine and bromine, requires careful handling due to their toxic and corrosive nature. Additionally, the generation of halogenated by-products, some of which may be environmentally persistent or harmful, necessitates consideration of green chemistry principles and mitigation strategies in industrial applications.
-
Initiation step of methane chlorination
-
Propagation step of methane chlorination
-
Termination step of methane chlorination
-
Methane radical chlorination
-
Toluene bromination
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian