Tin(II) chloride
Tin(II) chloride (also known as stannous chloride) is a white crystalline solid with the formula SnCl2. It forms a stable dihydrate, but aqueous solutions tend to undergo hydrolysis, particularly if hot. SnCl2 is widely used as a reducing agent (in acid solution), and in electrolytic baths for tin-plating. Tin(II) chloride should not be confused with the other chloride of tin; tin(IV) chloride or stannic chloride (SnCl4).
Properties[edit]
Tin(II) chloride is a strong reducing agent and is easily oxidized by atmospheric oxygen. Solid tin(II) chloride can be crystallized as a pentahydrate (SnCl2·5H2O), which is the form that is most commonly sold commercially. The anhydrous form is a crystalline solid that is colorless to slightly yellow.
Preparation[edit]
Tin(II) chloride can be prepared by the action of hydrochloric acid on tin, or by dissolving tin in concentrated hydrochloric acid and evaporating. It can also be prepared by the reaction of tin with chlorine gas.
Uses[edit]
Tin(II) chloride is used for tin-plating of steel, in the manufacture of tin cans, in textile dyeing as a mordant, and in making pigments. It is also used in the production of catalysts, used in the manufacture of polyurethane foams and high performance resins.
Safety[edit]
Tin(II) chloride is not considered highly toxic, but it is an irritant to the eyes and skin, and if ingested or inhaled in large amounts can cause nausea, vomiting, diarrhea and abdominal pain.
See also[edit]
References[edit]
<references />
Tin(II) chloride[edit]
-
Tin(II) chloride
-
Structure of SnCl2
-
3D ball model of Tin(II) chloride crystal
-
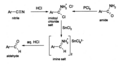
Stephen reduction using SnCl2
-
Nitro reduction scheme with SnCl2
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian