Iodine heptafluoride

Iodine heptafluoride, also known as iodine(VII) fluoride or IF7, is a chemical compound with the formula IF7. It is one of the interhalogen compounds, consisting of halogens iodine and fluorine. Iodine heptafluoride is notable for its unusual molecular geometry, known as a pentagonal bipyramid. It is a colorless, reactive gas that is used in some chemical syntheses and has been studied for various applications in chemistry.
Properties[edit]
Iodine heptafluoride is a colorless gas at room temperature, with a sharp, pungent odor. It is exceptionally reactive, particularly with water, where it hydrolyzes to form hydrofluoric acid (HF) and iodic acid (HIO3). IF7 is a strong oxidizing agent and can react explosively with organic materials or reducing agents.
Synthesis[edit]
Iodine heptafluoride can be synthesized by the direct reaction of iodine with fluorine gas at high temperatures. The reaction is highly exothermic and must be conducted with caution to avoid explosions:
\[I_2 + 7 F_2 \rightarrow 2 IF_7\]
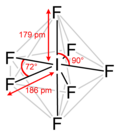
Structure[edit]
The molecular structure of IF7 is a pentagonal bipyramid. Each fluorine atom is equidistant from the central iodine atom, and the molecule exhibits D5h symmetry. This geometry is a result of the VSEPR theory, which predicts the shape of molecules based on the repulsion between electron pairs around the central atom.
Reactivity[edit]
Due to its high reactivity, IF7 is used in specialized chemical syntheses, such as the fluorination of certain organic compounds. Its reactivity with water and other nucleophiles is utilized in some industrial processes to produce fluorinated compounds.
Safety[edit]
Iodine heptafluoride is highly corrosive and toxic, requiring special handling and storage conditions. It can cause severe burns upon contact with skin and is dangerous if inhaled. Safety measures include the use of protective equipment and proper ventilation when handling the gas.
Applications[edit]
While the applications of IF7 are limited due to its reactivity and toxicity, it is used in certain areas of organic synthesis and industrial chemistry. Its ability to introduce fluorine atoms into organic molecules is valuable in the production of some pharmaceuticals and agrochemicals.
-
2D dimensions of Iodine heptafluoride
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian