Formaldehyde
[[File:||thumb]]

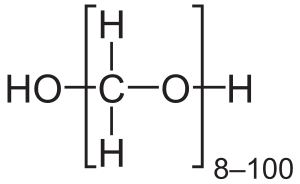


Formaldehyde is a naturally occurring organic compound with the formula CH2O (H-CHO). It is the simplest of the aldehydes (R-CHO) and is also known by its systematic name methanal. The common name of this substance comes from its similarity and relation to formic acid. Formaldehyde is a colorless gas with a pungent, distinct odor and is used in various chemical manufacturing processes, primarily in the production of industrial resins, such as urea-formaldehyde and phenol-formaldehyde resins.
Properties and Structure[edit]
Formaldehyde is a highly reactive, flammable gas at room temperature. It is soluble in water, alcohol, and other solvents. The structure of formaldehyde consists of a carbon atom double-bonded to an oxygen atom (carbonyl group) and two hydrogen atoms, making it the simplest member of the aldehyde group.
Production[edit]
Formaldehyde is primarily produced by the catalytic oxidation of methanol. The most common process involves methanol and air in the presence of a metal oxide catalyst. This process yields formaldehyde along with a small amount of formic acid.
Uses[edit]
Formaldehyde's primary use is in the production of industrial resins and plastics. For instance, urea-formaldehyde resins are used in the manufacture of particle board and other pressed wood products, while phenol-formaldehyde resins are essential in the production of insulation materials. Formaldehyde is also used as a preservative in some paints and coatings, and as a disinfectant and biocide in medical laboratories and other settings.
Health Concerns[edit]
Exposure to formaldehyde can cause various health issues. Short-term exposure to high levels of formaldehyde may lead to respiratory discomfort, skin irritation, and eye irritation. Long-term exposure has been associated with more severe health effects, including certain types of cancer. The International Agency for Research on Cancer (IARC) classifies formaldehyde as a Group 1 carcinogen, indicating that there is sufficient evidence that it can cause cancer in humans.
Regulation[edit]
Due to its potential health risks, the use and handling of formaldehyde are regulated in many countries. Regulations may include limits on workplace exposure levels, requirements for safety equipment and ventilation systems, and restrictions on the concentration of formaldehyde in consumer products.
Environmental Impact[edit]
Formaldehyde is also a significant concern in terms of environmental impact. It can be released into the air from industrial processes and burning of biomass and fossil fuels. Once in the atmosphere, formaldehyde acts as a volatile organic compound (VOC), contributing to the formation of ground-level ozone and smog, which have adverse effects on air quality and human health.
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
