Aromaticity



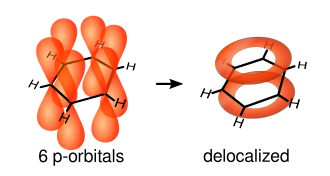

Aromaticity is a chemical property of conjugated ring systems that results in unusual stability and specific electronic configurations. The concept is fundamental in organic chemistry and plays a vital role in understanding the structure, reactivity, and properties of many organic molecules, ranging from simple benzene derivatives to complex heterocyclic compounds and polycyclic aromatic hydrocarbons.
Definition and Characteristics[edit]
Aromatic compounds are characterized by their ability to delocalize π electrons across the ring structure, following Hückel's rule, which states that a molecule is aromatic if it contains a planar ring of continuously overlapping p-orbitals with (4n+2) π electrons, where n is a non-negative integer. This delocalization leads to enhanced stability compared to non-aromatic compounds, making aromatic compounds less reactive in certain types of chemical reactions.
History[edit]
The concept of aromaticity was first introduced in the early 19th century with the discovery of benzene by Michael Faraday in 1825. The unique stability of benzene, despite its unsaturated nature, puzzled chemists until Friedrich August Kekulé proposed the cyclic structure with alternating single and double bonds in 1865. However, the true nature of benzene's stability and electron delocalization was not fully understood until the development of quantum chemistry in the 20th century.
Types of Aromatic Compounds[edit]
Aromatic compounds can be broadly classified into two categories: benzenoid aromatic compounds, which contain one or more benzene rings, and non-benzenoid aromatic compounds, which do not contain benzene rings but meet the criteria of aromaticity. Additionally, heterocyclic aromatic compounds contain at least one atom other than carbon within the ring, such as nitrogen, oxygen, or sulfur, contributing to the aromatic system.
Applications[edit]
Aromatic compounds have widespread applications in various fields, including pharmaceuticals, where many drugs contain aromatic rings due to their stability and reactivity. They are also used in the manufacture of dyes, plastics, synthetic fibers, and agrochemicals. In the field of organic electronics, aromatic compounds are key components due to their ability to conduct electricity.
Environmental and Health Concerns[edit]
Some aromatic compounds, particularly certain polycyclic aromatic hydrocarbons, are known to be toxic and carcinogenic, posing environmental and health risks. These compounds can be released into the environment through the incomplete combustion of organic matter and are the subject of ongoing research and regulation to mitigate their impact.
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
