Active site
Active Site
The active site of an enzyme is the region where substrate molecules bind and undergo a chemical reaction. The active site is typically a small pocket or groove on the surface of the enzyme, formed by the three-dimensional arrangement of amino acids. This site is crucial for the enzyme's catalytic activity and specificity.
Structure and Function
The active site is composed of residues that create a specific environment for the substrate. These residues can participate directly in the chemical transformation of the substrate or help position the substrate correctly. The active site is often highly specific to the substrate, a concept known as the lock and key model.

Induced Fit Model
The induced fit model suggests that the active site is flexible and can change shape to better fit the substrate upon binding. This model accounts for the dynamic nature of enzyme-substrate interactions.

Catalytic Mechanisms
Enzymes can employ various mechanisms to catalyze reactions, including:
- Acid-base catalysis: Involves the transfer of protons.
- Covalent catalysis: Involves the formation of a transient covalent bond between the enzyme and the substrate.
- Metal ion catalysis: Utilizes metal ions to stabilize charged intermediates.
- Proximity and orientation effects: Increases the effective concentration of substrates and aligns them for reaction.
Factors Affecting Active Site Function
Several factors can influence the activity of the active site, including:
- pH and temperature: These can affect the ionization state of the active site residues and the overall enzyme structure.
- Inhibitors: Molecules that can bind to the active site or elsewhere on the enzyme to reduce its activity.
- Cofactors and coenzymes: Non-protein molecules that assist in the catalytic activity of the enzyme.
Examples of Active Sites
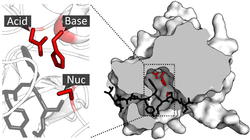
Catalytic Triad
A common feature in many proteases is the catalytic triad, which consists of three amino acids that work together to perform catalysis. An example is the triad found in TEV protease.
Redox Reactions
Enzymes like glutathione reductase use active sites to facilitate redox reactions, involving the transfer of electrons.

Related Pages
Gallery
-
Structure of an enzyme showing the active site.
-
Illustration of enzyme inhibition.
-
Hydrogen bonding in water, relevant to enzyme interactions.
-
Dipole-dipole interactions in enzyme-substrate binding.
-
Hydrophobic interactions in protein structure.
-
Mechanism of peptide bond cleavage in _-chymotrypsin.
-
Redox states of FAD, a cofactor in enzymatic reactions.
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
-
Active_site
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian











