Flosequinan: Difference between revisions
CSV import |
No edit summary |
||
| Line 21: | Line 21: | ||
{{medicine-stub}} | {{medicine-stub}} | ||
<gallery> | <gallery> | ||
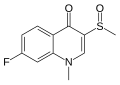
File:Flosequinan.svg|Flosequinan | File:Flosequinan.svg|Flosequinan | ||
</gallery> | </gallery> | ||
Latest revision as of 21:54, 16 March 2025
Flosequinan is a vasodilator that was once used in the treatment of congestive heart failure. It functions by relaxing the blood vessels, which reduces the workload on the heart, making it easier for the heart to pump blood throughout the body. Despite its initial promise, Flosequinan was withdrawn from the market due to concerns over its safety and an associated increase in mortality among patients.
Mechanism of Action[edit]
Flosequinan works by inhibiting phosphodiesterase enzymes, leading to an increase in intracellular cyclic guanosine monophosphate (cGMP). The increase in cGMP levels causes smooth muscle relaxation in the blood vessels, leading to vasodilation. This mechanism is similar to that of other vasodilators used in the management of heart failure and hypertension.
Clinical Uses[edit]
Initially, Flosequinan was approved for the management of congestive heart failure (CHF) to improve symptoms and increase exercise tolerance in patients. Its ability to dilate blood vessels and reduce the workload on the heart made it a valuable addition to heart failure treatment regimens at the time.
Adverse Effects[edit]
While Flosequinan was effective in relieving symptoms associated with heart failure, its use was associated with several adverse effects. These included headache, dizziness, and nausea, which are common to vasodilators. More concerning, however, was the increased risk of mortality observed in patients taking Flosequinan for extended periods, which ultimately led to its withdrawal from the market.
Market Withdrawal[edit]
The decision to withdraw Flosequinan from the market was based on data from long-term clinical trials that demonstrated an increased risk of mortality and hospitalization for patients with advanced heart failure. This led regulatory authorities to reassess the risk-benefit profile of the drug, concluding that the risks outweighed the benefits for patients with heart failure.
Conclusion[edit]
Flosequinan serves as an example of the complexities involved in drug development and the importance of long-term safety studies in the evaluation of new therapies. While it offered symptomatic relief for patients with congestive heart failure, the long-term risks associated with its use necessitated its withdrawal from clinical use.
-
Flosequinan