Arsenic trifluoride: Difference between revisions
CSV import |
CSV import |
||
| Line 28: | Line 28: | ||
{{Chem-stub}} | {{Chem-stub}} | ||
{{No image}} | {{No image}} | ||
== Arsenic trifluoride gallery == | |||
<gallery> | |||
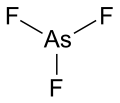
File:Arsenic(III)fluoride.svg|Arsenic(III)fluoride | |||
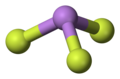
File:Arsenic-trifluoride-3D-balls.png|Arsenic trifluoride 3D balls | |||
</gallery> | |||
Revision as of 05:11, 3 March 2025
Arsenic trifluoride is a chemical compound with the formula AsF3. This inorganic compound is a colorless liquid and is one of the main fluorides of arsenic, the others being AsF5 and AsF. Arsenic trifluoride is used in the manufacturing of other arsenic compounds and in some chemical synthesis processes due to its reactivity.
Properties
Arsenic trifluoride is a colorless, poisonous, and volatile liquid. It has a pungent odor and is highly toxic, similar to other arsenic compounds. The compound has a molecular weight of 131.92 g/mol and boils at 63 °C. It is slightly soluble in water but reacts with it, releasing hydrogen fluoride (HF) and forming arsenous acid (H3AsO3).
Synthesis
Arsenic trifluoride can be synthesized by the reaction of arsenic or arsenic oxide with hydrogen fluoride. The process involves the direct combination of the elements under controlled conditions.
\[ \text{2 As + 6 HF} \rightarrow \text{2 AsF3 + 3 H2} \]
Alternatively,
\[ \text{As2O3 + 6 HF} \rightarrow \text{2 AsF3 + 3 H2O} \]
Applications
The primary use of arsenic trifluoride is in the synthesis of organic and inorganic arsenic compounds. It serves as a fluorinating agent in chemical reactions, where it introduces fluorine atoms into other molecules. Despite its toxicity, its reactivity is exploited in specialized organic synthesis processes.
Safety
Due to its high toxicity and reactivity, especially towards water and moisture, handling arsenic trifluoride requires strict safety precautions. Protective equipment, such as gloves and goggles, and adequate ventilation are essential when working with this compound. Ingestion, inhalation, or skin contact can lead to severe arsenic poisoning, and appropriate measures should be taken to prevent exposure.
Environmental Impact
Arsenic trifluoride, like other arsenic compounds, poses a significant environmental hazard. Its release into the environment can lead to contamination of water and soil, affecting wildlife and potentially entering the human food chain. Efforts to minimize its environmental impact include proper storage, handling, and disposal procedures to prevent leaks and spills.
Arsenic trifluoride gallery
-
Arsenic(III)fluoride
-
Arsenic trifluoride 3D balls