Haloalkane: Difference between revisions
CSV import |
CSV import |
||
| Line 32: | Line 32: | ||
{{stub}} | {{stub}} | ||
{{dictionary-stub1}} | {{dictionary-stub1}} | ||
<gallery> | |||
File:Tetrafluoroethane liquid.jpg|Haloalkane | |||
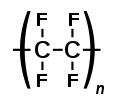
File:Polytetrafluoroethylene.svg|Haloalkane | |||
</gallery> | |||
Latest revision as of 01:47, 20 February 2025
Haloalkane or alkyl halides are a group of chemical compounds, consisting of alkanes, such as methane or ethane, with one or more halogens linked, such as chlorine or fluorine, making them a type of organic halide.
Structure and Physical Properties[edit]
Haloalkanes are classified into different types based on the type and arrangement of the halogen atom. They can be classified as primary, secondary or tertiary haloalkanes. The carbon-halogen bond in haloalkanes is polarized because of the difference in electronegativity between the carbon and the halogen atoms.
Synthesis[edit]
Haloalkanes can be synthesized from alcohols by using a halogen acid, phosphorus halides, or thionyl chloride. They can also be prepared from alkanes through free radical halogenation.
Reactions[edit]
Haloalkanes undergo various reactions, including nucleophilic substitution, elimination reactions, and reactions with metals.
Uses[edit]
Haloalkanes are used in a wide variety of applications, including refrigerants, aerosol propellants, solvents, and pharmaceuticals.
Health Effects[edit]
Exposure to haloalkanes can cause various health effects, including skin and eye irritation, respiratory problems, and in severe cases, damage to the liver and kidneys.
See Also[edit]
References[edit]
<references />