Ene reaction: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 32: | Line 32: | ||
{{Chemistry-stub}} | {{Chemistry-stub}} | ||
<gallery> | |||
File:Ene_reaction.svg|Ene reaction | |||
File:Ene_reaction_orbitals_and_transition_state.svg|Ene reaction orbitals and transition state | |||
File:Jatropha-5,12-diene_carbonyl-ene_reaction.svg|Jatropha-5,12-diene carbonyl-ene reaction | |||
File:Figure4ene.png|Ene reaction | |||
File:EneIsoprene.svg|Ene reaction | |||
File:Figure6ene.png|Ene reaction | |||
File:Figure7ene.png|Ene reaction | |||
File:Lewis_acid-catalyzed_ene_reaction.svg|Lewis acid-catalyzed ene reaction | |||
File:Figure9ene.png|Ene reaction | |||
File:Figure10ene.png|Ene reaction | |||
File:Figure11ene.png|Ene reaction | |||
File:Figure12ene.png|Ene reaction | |||
</gallery> | |||
Latest revision as of 11:51, 18 February 2025
Ene Reaction
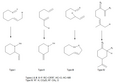
The Ene Reaction is a fundamental chemical reaction that involves the transfer of a hydrogen atom from one molecule (the ene) to an unsaturated species (the enophile) in the presence of a catalyst. This reaction is a key process in organic chemistry, particularly in the synthesis of complex natural products and the modification of unsaturated compounds. The Ene Reaction is characterized by its versatility and efficiency in forming carbon-carbon and carbon-heteroatom bonds, making it an essential tool in the arsenal of synthetic chemists.
Mechanism[edit]
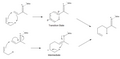
The Ene Reaction proceeds through a concerted mechanism, which involves the simultaneous formation and breaking of bonds. The reaction typically occurs through a six-membered transition state, where the hydrogen atom is transferred from the ene to the enophile, resulting in the formation of a new alkene and a new bond between the ene and enophile. The reaction can be catalyzed by both acids and bases, with the choice of catalyst affecting the reaction rate and selectivity.
Types of Ene Reactions[edit]
There are several variations of the Ene Reaction, each with its own specific substrates and conditions. These include:
- Thermal Ene Reactions: Occur without a catalyst at elevated temperatures. These reactions are typically less selective but can be useful for substrates that are stable to heat.
- Catalytic Ene Reactions: Involve the use of a catalyst, such as a Lewis acid or a Brønsted acid, to lower the activation energy and increase the reaction rate. Catalytic ene reactions are more selective and can be conducted at lower temperatures.
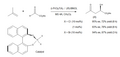
- Asymmetric Ene Reactions: A subset of catalytic ene reactions that employ chiral catalysts to produce enantiomerically enriched products. These reactions are important in the synthesis of chiral molecules.
Applications[edit]
The Ene Reaction finds widespread application in organic synthesis, including the synthesis of natural products, pharmaceuticals, and polymers. Its ability to efficiently create complex molecules from simpler precursors has made it a valuable tool in the development of new drugs and materials.
Limitations[edit]
Despite its versatility, the Ene Reaction has some limitations. The reaction's efficiency and selectivity can be affected by the nature of the substrates and the reaction conditions. Additionally, some substrates may undergo competing reactions, such as polymerization or rearrangement, which can complicate the reaction outcome.
See Also[edit]
References[edit]
<references/>
-
Ene reaction
-
Ene reaction orbitals and transition state
-
Jatropha-5,12-diene carbonyl-ene reaction
-
Ene reaction
-
Ene reaction
-
Ene reaction
-
Ene reaction
-
Lewis acid-catalyzed ene reaction
-
Ene reaction
-
Ene reaction
-
Ene reaction
-
Ene reaction