Fick's laws of diffusion: Difference between revisions
CSV import |
CSV import |
||
| Line 46: | Line 46: | ||
{{Chemistry-stub}} | {{Chemistry-stub}} | ||
{{Biology-stub}} | {{Biology-stub}} | ||
<gallery> | |||
File:DiffusionMicroMacro.gif|Fick's laws of diffusion | |||
File:Diffusive_sorption_probability.png|Diffusive sorption probability | |||
File:DiffusiveAdsorptionHistory.jpg|Diffusive adsorption history | |||
File:JChen2022JPCA.png|Fick's laws of diffusion | |||
</gallery> | |||
Latest revision as of 04:28, 18 February 2025
Fick's laws of diffusion describe the transport of particles or substances from areas of higher concentration to areas of lower concentration. Developed by Adolf Fick in 1855, these laws are fundamental in understanding processes such as gas exchange in the lungs, the diffusion of nutrients and waste products between blood and tissues, and the movement of substances across cell membranes.
Fick's First Law[edit]
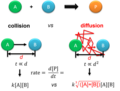
Fick's first law relates to the steady-state diffusion, which is the diffusion flux in systems not dependent on time. It states that the diffusion flux is proportional to the concentration gradient. Mathematically, it is expressed as:
\[ J = -D \frac{\partial \phi}{\partial x} \]
where:
- \(J\) is the diffusion flux, the amount of substance per unit area per unit time (e.g., \(mol \cdot m^{-2} \cdot s^{-1}\)).
- \(D\) is the diffusion coefficient or diffusivity of the substance in the medium (e.g., \(m^2 \cdot s^{-1}\)).
- \(\frac{\partial \phi}{\partial x}\) is the concentration gradient, the change in concentration with respect to distance.
The negative sign indicates that diffusion occurs in the direction opposite to the gradient.
Fick's Second Law[edit]
Fick's second law predicts how diffusion causes the concentration to change over time. It is derived from Fick's first law and the principle of conservation of mass. Fick's second law is given by:
\[ \frac{\partial \phi}{\partial t} = D \frac{\partial^2 \phi}{\partial x^2} \]
where:
- \(\frac{\partial \phi}{\partial t}\) is the rate of change of concentration with time.
- \(\frac{\partial^2 \phi}{\partial x^2}\) is the second derivative of concentration with respect to position, indicating how the concentration gradient changes in space.
This equation is used to model changing concentration profiles in a medium over time.
Applications[edit]
Fick's laws are applied in various fields, including:
- Biology, for understanding processes such as osmosis, respiration, and cellular diffusion.
- Chemistry, in the study of reaction kinetics and the mixing of chemicals.
- Physics and engineering, for solving problems related to heat transfer and material properties.
Limitations[edit]
While Fick's laws provide a foundation for understanding diffusion processes, they have limitations. They assume a homogeneous and isotropic medium, constant diffusivity, and neglect convective movements that can occur in fluids. In complex systems, such as biological tissues or porous materials, these assumptions may not hold true.
See Also[edit]

-
Fick's laws of diffusion
-
Diffusive sorption probability
-
Diffusive adsorption history
-
Fick's laws of diffusion