Phthalein dye: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import Tags: mobile edit mobile web edit |
||
| Line 1: | Line 1: | ||
{{DISPLAYTITLE:Phthalein Dye}} | |||
== | == Phthalein Dye == | ||
[[File:Phenolphthalein.svg|thumb|right|Structural formula of phenolphthalein]] | |||
Phthalein dyes are a class of synthetic dyes derived from [[phthalic anhydride]] and phenols. These dyes are known for their vibrant colors and are used in various applications, including as [[pH indicator]]s in [[chemistry]]. One of the most well-known phthalein dyes is [[phenolphthalein]], which is commonly used in titrations. | |||
Phthalein dyes are used in | |||
== | == Structure and Synthesis == | ||
Phthalein dyes are characterized by their [[lactone]] ring structure, which is formed by the reaction of phthalic anhydride with phenols. The general reaction involves the condensation of phthalic anhydride with two equivalents of a phenol, resulting in the formation of a phthalein dye. | |||
The structure of phenolphthalein, a typical phthalein dye, consists of a central lactone ring connected to two phenolic groups. This structure is responsible for its color-changing properties in different pH environments. | |||
== | == Properties == | ||
Phthalein dyes | |||
Phthalein dyes exhibit a range of colors depending on their chemical structure and the pH of the solution they are in. For example, phenolphthalein is colorless in acidic solutions but turns pink to fuchsia in basic solutions. This property makes it an excellent pH indicator. | |||
The color change occurs due to the opening of the lactone ring in basic conditions, which alters the electronic structure of the dye and results in a visible color change. | |||
== Applications == | |||
Phthalein dyes are used in various applications: | |||
* '''pH Indicators''': Phenolphthalein is widely used in [[acid-base titration]]s to determine the endpoint of a reaction. | |||
* '''Dyeing''': Some phthalein dyes are used in the textile industry for dyeing fabrics. | |||
* '''Biological Staining''': Certain phthalein dyes are used in [[histology]] and [[cytology]] for staining tissues and cells. | |||
== Related Pages == | |||
* [[Phenolphthalein]] | |||
* [[pH Indicator]] | |||
* [[Acid-base titration]] | |||
* [[Dye]] | |||
[[Category:Dyes]] | [[Category:Dyes]] | ||
[[Category: | [[Category:Chemical indicators]] | ||
Latest revision as of 10:57, 15 February 2025
Phthalein Dye[edit]
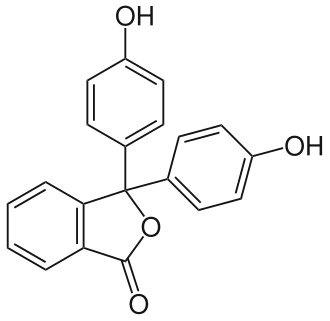
Phthalein dyes are a class of synthetic dyes derived from phthalic anhydride and phenols. These dyes are known for their vibrant colors and are used in various applications, including as pH indicators in chemistry. One of the most well-known phthalein dyes is phenolphthalein, which is commonly used in titrations.
Structure and Synthesis[edit]
Phthalein dyes are characterized by their lactone ring structure, which is formed by the reaction of phthalic anhydride with phenols. The general reaction involves the condensation of phthalic anhydride with two equivalents of a phenol, resulting in the formation of a phthalein dye.
The structure of phenolphthalein, a typical phthalein dye, consists of a central lactone ring connected to two phenolic groups. This structure is responsible for its color-changing properties in different pH environments.
Properties[edit]
Phthalein dyes exhibit a range of colors depending on their chemical structure and the pH of the solution they are in. For example, phenolphthalein is colorless in acidic solutions but turns pink to fuchsia in basic solutions. This property makes it an excellent pH indicator.
The color change occurs due to the opening of the lactone ring in basic conditions, which alters the electronic structure of the dye and results in a visible color change.
Applications[edit]
Phthalein dyes are used in various applications:
- pH Indicators: Phenolphthalein is widely used in acid-base titrations to determine the endpoint of a reaction.
- Dyeing: Some phthalein dyes are used in the textile industry for dyeing fabrics.
- Biological Staining: Certain phthalein dyes are used in histology and cytology for staining tissues and cells.