Sulfur: Difference between revisions
CSV import |
CSV import |
||
| Line 32: | Line 32: | ||
{{stub}} | {{stub}} | ||
{{dictionary-stub1}} | {{dictionary-stub1}} | ||
<gallery> | |||
File:Burning-sulfur.png|Sulfur | |||
File:Liquid_hydrogen_sulfide.png|Sulfur | |||
File:Sulphuric_acid_96_percent_extra_pure.jpg|Sulfur | |||
File:Cyclooctasulfur-above-3D-balls.png|Sulfur | |||
File:Nearly_exhausted_sulphur_vat_from_which_railroad_cars_are_loaded,_Freeport_Sulphur_Co.,_Hoskins_Mound,_Texas,_1a35438v.jpg|Sulfur | |||
File:Io_highest_resolution_true_color.jpg|Sulfur | |||
File:Menambang_Belerang.jpg|Sulfur | |||
File:Kawah_Ijen_-East_Java_-Indonesia_-sulphur-31July2009.jpg|Sulfur | |||
File:Lapis_lazuli_block.jpg|Sulfur | |||
File:S@CNT.jpg|Sulfur | |||
File:L-Cystein_-_L-Cysteine.svg|Sulfur | |||
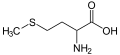
File:Methionin_-_Methionine.svg|Sulfur | |||
</gallery> | |||
Revision as of 11:52, 18 February 2025
Sulfur or sulphur is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature.
Characteristics
Sulfur is a bright yellow crystalline solid. It is characterized by its odorous nature, often associated with the smell of rotten eggs, due to the presence of sulfur compounds. Sulfur is multivalent, meaning it has several common oxidation states, including -2, +2, +4 and +6.
Occurrence
Sulfur occurs naturally as the pure element (native sulfur) and as sulfide and sulfate minerals. It is an essential element for life and is found in two amino acids, cysteine and methionine.
Uses
Sulfur is used in many industries, including the production of sulfuric acid, the world's most produced chemical. It is also used in the vulcanization of rubber, as a fungicide, and as a component in black gunpowder.
History
The history of sulfur is rooted in antiquity, with mentions of the element found in the Biblical texts. It was known to the ancients in its native form and was also part of the primitive gunpowder formulation.
See also
References
<references />