Combretastatin A-4: Difference between revisions
CSV import |
CSV import Tags: mobile edit mobile web edit |
||
| Line 1: | Line 1: | ||
{{DISPLAYTITLE:Combretastatin A-4}} | |||
== | == Overview == | ||
Combretastatin A-4 is a | [[File:Combretastatin_A4.svg|thumb|right|Chemical structure of Combretastatin A-4]] | ||
'''Combretastatin A-4''' is a naturally occurring [[stilbenoid]] that is derived from the bark of the South African tree ''[[Combretum caffrum]]''. It is a potent [[antineoplastic]] agent that has been studied for its ability to disrupt the [[microtubule]] network in [[cancer]] cells, leading to [[apoptosis]]. | |||
== | == Chemical Structure == | ||
Combretastatin A-4 is characterized by its [[cis]]-stilbene structure, which is crucial for its biological activity. The molecule consists of two aromatic rings connected by a double bond, with various substituents that enhance its binding to the [[tubulin]] protein. | |||
== Mechanism of Action == | |||
Combretastatin A-4 exerts its effects by binding to the [[colchicine]] site on [[tubulin]], a protein that is essential for the formation of [[microtubules]]. By inhibiting tubulin polymerization, Combretastatin A-4 disrupts the [[mitotic spindle]] formation, leading to cell cycle arrest and subsequent apoptosis in rapidly dividing cancer cells. | |||
==Clinical | == Clinical Applications == | ||
While Combretastatin A-4 itself has not been widely used in clinical settings, its derivatives, such as [[fosbretabulin]] (also known as CA4P), have been investigated in clinical trials for the treatment of various [[solid tumors]]. These compounds are often used in combination with other [[chemotherapeutic agents]] to enhance their efficacy. | |||
== | == Research and Development == | ||
Ongoing research is focused on improving the pharmacokinetic properties of Combretastatin A-4 and its derivatives. Efforts are being made to enhance their [[bioavailability]] and reduce potential [[toxicity]]. Additionally, studies are exploring the use of Combretastatin A-4 in combination with other [[targeted therapies]] to overcome [[drug resistance]] in cancer treatment. | |||
== | == Related Pages == | ||
* [[Stilbenoid]] | |||
* [[Microtubule]] | |||
* [[Apoptosis]] | |||
* [[Tubulin]] | |||
* [[Colchicine]] | |||
* [[Fosbretabulin]] | |||
[[Category:Antineoplastic agents]] | [[Category:Antineoplastic agents]] | ||
[[Category:Stilbenoids]] | [[Category:Stilbenoids]] | ||
[[Category: | [[Category:Microtubule inhibitors]] | ||
Latest revision as of 11:13, 15 February 2025
Overview[edit]
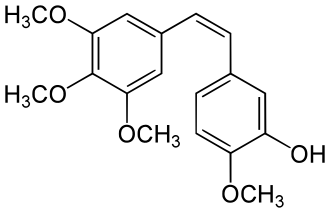
Combretastatin A-4 is a naturally occurring stilbenoid that is derived from the bark of the South African tree Combretum caffrum. It is a potent antineoplastic agent that has been studied for its ability to disrupt the microtubule network in cancer cells, leading to apoptosis.
Chemical Structure[edit]
Combretastatin A-4 is characterized by its cis-stilbene structure, which is crucial for its biological activity. The molecule consists of two aromatic rings connected by a double bond, with various substituents that enhance its binding to the tubulin protein.
Mechanism of Action[edit]
Combretastatin A-4 exerts its effects by binding to the colchicine site on tubulin, a protein that is essential for the formation of microtubules. By inhibiting tubulin polymerization, Combretastatin A-4 disrupts the mitotic spindle formation, leading to cell cycle arrest and subsequent apoptosis in rapidly dividing cancer cells.
Clinical Applications[edit]
While Combretastatin A-4 itself has not been widely used in clinical settings, its derivatives, such as fosbretabulin (also known as CA4P), have been investigated in clinical trials for the treatment of various solid tumors. These compounds are often used in combination with other chemotherapeutic agents to enhance their efficacy.
Research and Development[edit]
Ongoing research is focused on improving the pharmacokinetic properties of Combretastatin A-4 and its derivatives. Efforts are being made to enhance their bioavailability and reduce potential toxicity. Additionally, studies are exploring the use of Combretastatin A-4 in combination with other targeted therapies to overcome drug resistance in cancer treatment.