Thiocyanate: Difference between revisions
CSV import |
CSV import |
||
| Line 23: | Line 23: | ||
{{stub}} | {{stub}} | ||
<gallery> | |||
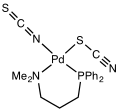
File:PalenikPdPN_SCN_NCSic1970.svg|Thiocyanate | |||
File:Pentaaquathiocyanatoiron(II)-3D-balls.png|Pentaaquathiocyanatoiron(II) 3D model | |||
File:Aqueous_ferric_thiocyanate_(Fe(SCN)n)_hydrate_mix.jpg|Aqueous ferric thiocyanate | |||
</gallery> | |||
Latest revision as of 04:13, 18 February 2025
Thiocyanate (also known as sulfocyanate) is a anion derived from the pseudohalogen compound hydrogen thiocyanate. It is a common component in pyrotechnics and is used in the production of certain herbicides and fungicides. Thiocyanate is also a byproduct of the metabolism of cysteine in the body.
Chemical Structure[edit]
Thiocyanate is composed of a sulfur atom, a carbon atom, and a nitrogen atom. The sulfur atom is bonded to the carbon atom, which is in turn bonded to the nitrogen atom. This linear structure is typical of pseudohalogen compounds.
Properties[edit]
Thiocyanate is a colorless, crystalline solid that is soluble in water. It has a melting point of 149°C and a boiling point of 335°C. It is a weak base, with a pKa of 1.3.
Uses[edit]
Thiocyanate is used in a variety of applications. It is a common component in pyrotechnics, where it is used to produce a red color. It is also used in the production of certain herbicides and fungicides. In the body, thiocyanate is a byproduct of the metabolism of cysteine.
Health Effects[edit]
Exposure to thiocyanate can have a variety of health effects. Ingestion can cause nausea, vomiting, and abdominal pain. Inhalation can cause coughing, shortness of breath, and chest pain. Long-term exposure can lead to thyroid problems, as thiocyanate can interfere with the uptake of iodine by the thyroid gland.
See Also[edit]
|
|
|
-
Thiocyanate
-
Pentaaquathiocyanatoiron(II) 3D model
-
Aqueous ferric thiocyanate