Galvanic corrosion: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 36: | Line 36: | ||
[[Category:Electrochemistry]] | [[Category:Electrochemistry]] | ||
[[Category:Metallurgy]] | [[Category:Metallurgy]] | ||
<gallery> | |||
File:Corrosão_de_ferro_por_sacrifício_em_contacto_com_cobre.jpg|Galvanic corrosion of iron in contact with copper | |||
File:Statue_lib_corr1.png|Galvanic corrosion on the Statue of Liberty | |||
File:Statue-de-la-liberte-new-york.jpg|Statue of Liberty | |||
File:Anodes-on-jacket.jpg|Anodes on a jacket structure | |||
File:Cathodic_Protection_Rectifier.JPG|Cathodic protection rectifier | |||
File:stainless-steel-mild-steel.jpg|Galvanic corrosion between stainless steel and mild steel | |||
File:Anode_sacrificielle.jpg|Sacrificial anode | |||
</gallery> | |||
Latest revision as of 11:18, 18 February 2025
Galvanic corrosion (also known as bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. This type of corrosion is characterized by the acceleration or deceleration of corrosive activity on a metal surface due to its proximity to another metal surface in a shared electrolyte environment.
Etymology[edit]
The term "galvanic" refers to Luigi Galvani, an Italian scientist who discovered in 1780 that connecting the muscle of a dead frog to two different metals caused the muscle to twitch. This phenomenon, later known as galvanism, is the basis for the naming of galvanic corrosion.
Mechanism[edit]
Galvanic corrosion occurs when two different metals have a physical or electrical contact with each other and are immersed in a common electrolyte, or when the same metal is exposed to electrolyte with different concentrations. In a galvanic couple, the more active metal (the anode) corrodes at an accelerated rate and the less active metal (the cathode) corrodes at a retarded rate. When a galvanic couple forms, one of the metals in the couple becomes the anode and corrodes faster than it would alone, while the other becomes the cathode and corrodes slower than it would alone.
Prevention[edit]
Galvanic corrosion can be prevented by:
- Selecting materials with similar corrosion potentials.
- Avoiding direct metal-to-metal contact.
- Applying protective coatings.
- Using suitable gaskets or insulators to separate the metals.
- Using cathodic protection.
Related terms[edit]
See also[edit]
References[edit]
<references />
|
|
|
-
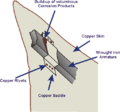
Galvanic corrosion of iron in contact with copper
-
Galvanic corrosion on the Statue of Liberty
-
Statue of Liberty
-
Anodes on a jacket structure
-
Cathodic protection rectifier
-
Galvanic corrosion between stainless steel and mild steel
-
Sacrificial anode