Coumarin: Difference between revisions
CSV import |
No edit summary |
||
| (2 intermediate revisions by the same user not shown) | |||
| Line 45: | Line 45: | ||
{{medicine-stub}} | {{medicine-stub}} | ||
<gallery> | |||
File:Coumarin_acsv.svg|Coumarin acsv | |||
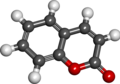
File:Coumarin.png|Coumarin | |||
File:Coumarin_spacefill.png|Coumarin spacefill | |||
</gallery> | |||
Latest revision as of 20:15, 16 March 2025
Coumarin is a fragrant organic chemical compound in the benzopyrone chemical class. It is a colorless crystalline substance in its standard state. Coumarin is found naturally in many plants, particularly in high concentration in the tonka bean, vanilla grass, sweet woodruff, and sweet clover. It has a sweet odor, readily recognized as the scent of newly-mown hay, and has been used in perfumes since 1882.
Chemical Structure and Properties[edit]
Coumarin has the chemical formula C_9H_6O_2. It consists of a benzene ring fused to a pyrone ring. The structure of coumarin is characterized by a lactone ring, which is a cyclic ester. This structure is responsible for its aromatic properties.
Natural Occurrence[edit]
Coumarin is found in a variety of plants, including:
These plants use coumarin as a defense mechanism against herbivores due to its toxic properties in high concentrations.
Uses[edit]
Coumarin has several applications:
- **Perfumery**: Due to its pleasant scent, coumarin is widely used in the fragrance industry.
- **Flavoring**: Although banned as a food additive in some countries due to its potential toxicity, coumarin is used in flavoring tobacco and certain alcoholic beverages.
- **Pharmaceuticals**: Coumarin derivatives, such as warfarin, are used as anticoagulants in medicine.
Health Effects[edit]
Coumarin can be toxic to the liver and kidneys if ingested in large quantities. It is metabolized in the liver to form 7-hydroxycoumarin, which is then excreted in the urine. Due to its potential toxicity, the use of coumarin as a food additive is regulated in many countries.
Related Compounds[edit]
Coumarin is related to several other compounds, including:
See Also[edit]
References[edit]
<references group="" responsive="1"></references>
External Links[edit]
-
Coumarin acsv
-
Coumarin
-
Coumarin spacefill