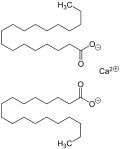
Calcium stearate
Calcium stearate is a carboxylate of calcium, classified as a type of fatty acid. It is a compound with a wide range of uses, including as a stabilizer in plastics, a lubricant, in the production of waterproofing materials, and in food production as an anti-caking agent.
Chemical Structure[edit]
Calcium stearate is composed of two stearate groups, or stearic acid molecules, and one calcium ion. In its pure form, it is a white, waxy powder. It is insoluble in water, but can be dissolved in hot pyridine. The compound can be obtained by the reaction of calcium chloride and sodium stearate in aqueous solution.
Uses[edit]
In Plastics[edit]
Calcium stearate is used as a stabilizer for PVC, and can also be used in the production of other types of plastics. It helps to prevent the breakdown of the plastic's molecular structure during processing, and also acts as a lubricant, reducing friction between the plastic and the machinery used to shape it.
In Food Production[edit]
In food production, calcium stearate is used as an anti-caking agent. It prevents ingredients from clumping together, which can affect the texture and appearance of the final product. It is generally considered safe for consumption, but is not suitable for people with a sensitivity to calcium or stearic acid.
Health and Safety[edit]
While calcium stearate is generally considered safe for use in food and in products that come into contact with the skin, it can cause irritation if inhaled. It is recommended that people working with the compound wear protective clothing and use appropriate ventilation.
See Also[edit]
-
Stearic Acid Calcium Salt Structural Formula
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian