Uric acid
A chemical compound and a product of purine metabolism
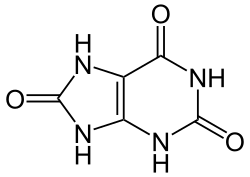
Uric acid is a heterocyclic compound of carbon, nitrogen, oxygen, and hydrogen with the formula C_H_N_O_. It forms ions and salts known as urates and acid urates, such as ammonium acid urate. Uric acid is a product of the metabolic breakdown of purine nucleotides, and it is a normal component of urine.
Structure and properties[edit]
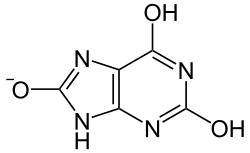
Uric acid is a weak acid and exists in several tautomeric forms, the most common being the keto form. It is a diprotic acid with pKa values of 5.4 and 10.3, meaning it can lose two protons. In its deprotonated form, it is known as urate.
Biological role[edit]
Uric acid is the end product of purine metabolism in humans and some other animals. In most mammals, uric acid is further oxidized by the enzyme uricase to allantoin, but in humans, this enzyme is not functional due to a mutation, leading to higher levels of uric acid in the blood.
Clinical significance[edit]
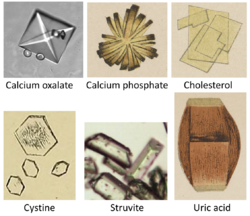
Elevated levels of uric acid in the blood, a condition known as hyperuricemia, can lead to the formation of urate crystals. These crystals can deposit in joints, causing gout, a form of inflammatory arthritis. Hyperuricemia is also associated with other medical conditions, such as kidney stones and cardiovascular disease.
Excretion[edit]
Uric acid is excreted by the kidneys in urine. The solubility of uric acid in urine is limited, and when the concentration exceeds its solubility, it can precipitate as crystals. These crystals can be seen in urine samples and are often used as a diagnostic tool.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian