6-APT
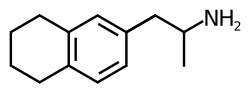
6-APT (6-aminopurine-2-thiol) is a chemical compound that belongs to the class of purine derivatives. It is structurally related to adenine, one of the four nucleobases in the nucleic acids of DNA and RNA.
Chemical Structure and Properties[edit]
6-APT is characterized by the presence of an amino group at the 6th position and a thiol group at the 2nd position of the purine ring. The molecular formula of 6-APT is C5H5N5S. The compound exhibits properties typical of purine derivatives, including the ability to form hydrogen bonds and participate in various biochemical reactions.
Synthesis[edit]
The synthesis of 6-APT can be achieved through several chemical pathways, often involving the modification of existing purine compounds. One common method involves the substitution of the hydrogen atom at the 6th position of adenine with an amino group, followed by the introduction of a thiol group at the 2nd position.
Applications[edit]
6-APT has been studied for its potential applications in biochemistry and pharmacology. It has been investigated for its role in enzyme inhibition, nucleic acid interactions, and as a potential therapeutic agent. Research is ongoing to explore its efficacy and safety in various medical and scientific contexts.
Biological Activity[edit]
The biological activity of 6-APT is primarily related to its interaction with nucleic acids and enzymes. It has been shown to inhibit certain enzymes involved in nucleic acid metabolism, which could have implications for the development of new antiviral and anticancer therapies.
Safety and Toxicity[edit]
As with many chemical compounds, the safety and toxicity of 6-APT depend on the dosage and exposure duration. Studies are required to fully understand its toxicological profile and to establish safe handling practices for laboratory and potential clinical use.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian