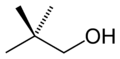
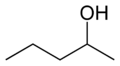
Amyl alcohol
Amyl alcohol (also known as pentanol) is a group of organic compounds with the formula C5H11OH. These compounds are primarily derived from natural fats and oils. They are colorless, liquid alcohols that are used in a variety of industrial applications.
Chemical Structure[edit]
Amyl alcohols have the general formula C5H11OH. This means they have five carbon atoms (C), eleven hydrogen atoms (H), and one hydroxyl group (-OH). The hydroxyl group is what classifies these compounds as alcohols.
Types of Amyl Alcohol[edit]
There are eight isomers of amyl alcohol, including:
- 1-Pentanol (n-pentanol, n-amyl alcohol)
- 2-Pentanol (sec-amyl alcohol)
- 3-Pentanol
- 2-Methyl-1-butanol (isopentyl alcohol)
- 3-Methyl-1-butanol (isoamyl alcohol)
- 2-Methyl-2-butanol (tert-amyl alcohol)
- 3-Methyl-2-butanol
- 2,2-Dimethyl-1-propanol
Uses[edit]
Amyl alcohols are used in a variety of industrial applications. They are used as solvents in the production of other chemicals, and as ingredients in perfumes and other fragrances. They are also used in the production of pharmaceuticals, and in the food and beverage industry as flavoring agents.
Health Effects[edit]
Exposure to amyl alcohols can cause a variety of health effects. Inhalation can cause respiratory irritation, dizziness, and nausea. Ingestion can cause gastrointestinal irritation, vomiting, and diarrhea. Long-term exposure can cause damage to the liver and kidneys.
See Also[edit]
References[edit]
<references />
-
Pentan-1-ol
-
2-Methyl-1-butanol
-
Isoamyl alcohol
-
Neopentyl alcohol
-
Pentan-2-ol
-
3-Methylbutan-2-ol
-
Pentan-3-ol
-
Tert-pentyl alcohol
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian